IJCRR - 11(6), March, 2019
Pages: 08-15
Date of Publication: 30-Mar-2019
Print Article
Download XML Download PDF
Seasonal and Geographical Variations in Antimicrobial Activity of Selected Mangroves from Krishna Estuary
Author: Vijaya Kumar Kovvada, Rosaiah Gorrepati, Babu Kakumanu, Tirupathi Swamy Nattala, Rajesh Butti
Category: Life Sciences
Abstract:Objective: To determine the seasonal and geographical impact on plant secondary metabolite production and antimicrobial activity.
Methods: Three mangrove plants viz., Ceriops tagal, Bruguiera cylindrica and Lumnitzera racemosa commonly found in Gilakaladindi and Malakayalanka regions of Krishna Estuary, Andhra Pradesh, India were selected for the present study. The study was done during 2014-15 summer, rainy and winter in both the stations. Leaf samples were extracted with methanol and subjected to preliminary phytochemical screening. The methanolic leaf extracts were tested against bacterial pathogens Bacillus cereus, B. subtilis, Staphylococcus aureus, Escherichia coli and a fungal pathogen Candida albicans.
Results: The methanolic extracts of leaves collected during rainy season showed maximum zone of inhibition at both the stations Gilakaladindi and Malakayalanka. Among the studied plants leaf methanolic extract of C.tagal was recorded maximum antibacterial activity against B. subtilis and B. cereus (18\?0.05 mm), followed by Bruguiera cylindrica both at Gilakaladindi and Malakayalanka respectively. But the extracts of L.racemosa showed less inhibition activity (7\?0.00 mm) in all the seasons at both the stations.
Conclusion: The study demonstrated that mangroves produce bio-active compounds optimally in rainy season that are effective against microbial proliferation which may further help in synthesis of natural antibiotics in place of commercial antibiotics to heal infectious diseases.
Keywords: Gilakaladindi, Malakayalanka, Phytochemicals, Rainy
Full Text:
INTRODUCTION
Mangroves are a specific group of plant communities, habituated to adverse environments includes high salt content, relative humidity with fluctuated water currents.[1] These contrary conditions consequently results in reduced photosynthetic rate and thereby reduced growth.[2,3] Of all the stressors soil and water salinity inflict more on plant development especially in semiarid and arid regions.[4] In order to cope up with these unfavorable conditions, mangrove switch on to different modifications at morphological, physiological, biochemical and molecular levels.[5] Production of secondary metabolites is one of such physiological change which tends the plant to protect themselves against harmful microorganisms along with grazing threat.[6] Bryant et al.[7] have hypothesized that when plants are stressed, an exchange occurs between carbon to biomass production or formation of defensive secondary compounds.
The local geo-climate, seasonal changes, external factors such as light, temperature and humidity may affect composition of secondary metabolites. The synthesis of secondary metabolites in response to environmental factors is nothing but an adoptable strategy leading to tolerance to abiotic stress.[8,9] On the other side the mangrove plant extracts have proven activity against human, animal and plant pathogens and have been used in folklore medicine since ages.[10,11,12] Till now more than 200 bio-active compounds have been isolated from true mangroves of tropical and subtropical population.[13] As said, seasonal changes influence much on production of secondary metabolites which is less explored and badly overlooked. On the other side right season for the collection of leaf material to extract maximum secondary metabolites is inevitable in order to get much benefit. Krishna estuary is one such neglected area with less exploration specifically on seasonal changes. The Krishna deltaic region is in subtropical humid climate with hot summers and moderate winters, which get annual rainfall due to south west monsoon.[14,15] Gilakaladindi and Malakayalanka are the prominent biodiverse mangrove reserves in Krishna estuary on East Coast of India located at latitudes 16° 8?56.1624??, 15°58?34.5354 and longitudes 81°9?46.029??, 81°5?53.3616?? respectively. Coupled with their richness and diversity mangroves of Gilakaladindi and Malakayalanka have also physiological peculiarities as a result of environmental conditions to which they belong. Each species of mangroves has a particular range of tolerance to environmental factors.[16, 17]
The present study aims at identify the seasonal and geo-climatic changes on secondary metabolite production in terms of antimicrobial activity on selected mangrove species Lumnitzera racemosa, Ceriops tagal and Bruguiera cylindrica.
MATERIALS AND METHODS
Collection of Plant Material
Leaf samples were collected during summer (March to May) rainy (July to September) coinciding with South west monsoons and in winter season (October to February). Two different sites were chosen and the distance between the sites was about 25 kms. Healthy and fresh leaves were collected from selected three mangroves i.e. Lumnitzera racemosa, Ceriops tagal and Bruguiera cylindrica of Krishna estuary. The collected leaves were washed with tap water and double distilled water until dust is removed from surface of the leaves. The leaves are shade dried at room temperature. Dry leaf material was powdered with the help of mechanical grinder and sieved.
Extraction
The leaf powders were extracted with 80% methanol using soxhlet apparatus for about 18 hours. The crude extracts obtained were evaporated and concentrated by using vacuum rotary evaporator (Buchi Labortech Ag, model I, R-215) under reduced pressure. The dried extracts were preserved at 4°C until further use.
Antimicrobial screening
The antibacterial activity of the obtained leaf extracts was carried out by agar well diffusion method. Nutrient agar (NA) was used for culturing the test bacteria. Nutreint agar medium was sterilized at 15 lbs pressure (121 °C) for 15 min, later cooled and inoculated with 0.1 ml of test bacterial suspension. The inoculated medium was poured into petri plates under aseptic conditions. After solidification, wells of about 5 mm diameter were made with sterilized cork borer [18]. Solvent extract (50 μl, 100 μl and 150 μl) was added to each well and the addition of solvent alone served as control. The inoculated plates were incubated at 30 °C and the diameter of the inhibition zone was measured after 24 h on bacterial pathogens such as Staphylococcus aureus (MTCC 3160), Bacillus subtilis (ATCC 6633), Bacillus cereus (MTCC 430) Escherichia coli (MTCC 43) and fungal pathogen Candida albicans (ATCC 10231).
Phytochemical screening
Shade dried plant material was extracted with 80% methanol and were tested for qualitative phytochemical screening. [19,20]
Alkaloids
The methanolic extract was evaporated to dryness and the residue is dissolved in 1% HCl. To this solution Mayer’s reagent was added. Appearance of any precipitate or turbidity indicates the presence of alkaloids.
Saponins
The plant extract is evaporated to dryness. Tap-water was added and shaken vigorously. Formation of persistent foam of about 2 cm is taken as a positive reaction.
Terpenoids and steroids
A 50% H2SO4 is added along the sides of the test tube containing a mixture of methanolic HCl and acetic anhydride. If there is any change in color, from green to blue-green (sometimes via red or blue) indicates the presence of terpenoids and steroids.
Tannin
The methanolic extract is evaporated to dryness and the residue was dissolved in water and tested with 1% gelatin solution and 1% gelatin salt solution (1 g) gelatin dissolved in 10 g of NaCl (w/w) to separate volumes. The appearance of white precipitate will be regarded as a positive reaction.
Anthocyanin
To the plant extract was added equal volume of methanolic HCl. Appearance of red or purple color indicates the presence of anthocyanidins.
Phenolic compounds
The formation of intense color in the extract, on adding 1-2 drops of 1% ferric chloride to the extract is considered as a positive reaction test.
Flavonoids
Few ml of methanolic extract is added with conc. HCl and Mg powder. The presence of flavonoids can be identified by the development of pink or magenta or red colored foam.
Coumarin
To the methanolic extract, a few drops of alcoholic sodium hydroxide were added. Formation of yellow color indicated the presence of coumarins.
Quinones
To the 1 ml of methanolic extract, 1 ml of conc. H2SO4 was added. Formation of red color shows the presence of Quinone’s.
Resins
Plant extracts were treated with acetone. To this, small amount of water was added and shaken. The appearance of turbidity indicates the presence of resins.
Test for glycosides
To the methanolic extract a little amount of anthrone was added. To this a few drops of conc. H2SO4 was added and warmed gently over water bath. The presence of glycosides was identified by dark green color formation.
Soil and Water analysis
Field data like temperature, salinity and pH were recorded during morning to noon. Water salinity was estimated with the help of hand refractometer and pH was measured using Elico pH meter (model L.C-120). Data was collected on five plants of each species growing in different locations in both the sites. Soils are air-dried, crushed with a wooden mallet, passed through a 2 mm sieve and were analyzed for inorganic ions using the standard procedure.[21] Saturation extracts of soils are analyzed for electrical conductivity using an electrical conductivity meter. Soil pH was determined on air-dried samples using a 1: 2.5 soil to 1 M KCl ratio. Exchangeable Na+ Ca2+, Mg2+, K+, P, Zn2+, Cu2+ and Mg2+ present soil and water samples were analyzed. [22]
RESULTS
The methanolic leaf extracts were screened for various phytochemical constituents and represented in Table 1. The Phytochemical analysis of the methanol extracts revealed the presence of alkaloids, flavanoids, phenols, resins, saponins and tannins in all the studied plants of both Gilakaladindi and Malakayalanka estuaries in all the seasons (Table 1). But the existence of anthocyanins, quinines, glycosides and terpenoids were not reported in L. racemosa in all the seasons at both the stations. Along with the L. racemosa the presence of anthocyanins and quinines not observed in B. cylindrica in all the seasons. The existence of flavonoids also not recorded in B. cylindrica in all the seasons at both the stations. On the other side the presence of coumarins and glycosides not observed in C. tagal in both the stations during all the seasons. These variations in presence or absence of phytochemicals among three selected mangroves may correspond to different bioactive constituents and may be responsible for variations in antimicrobial potency among the species.
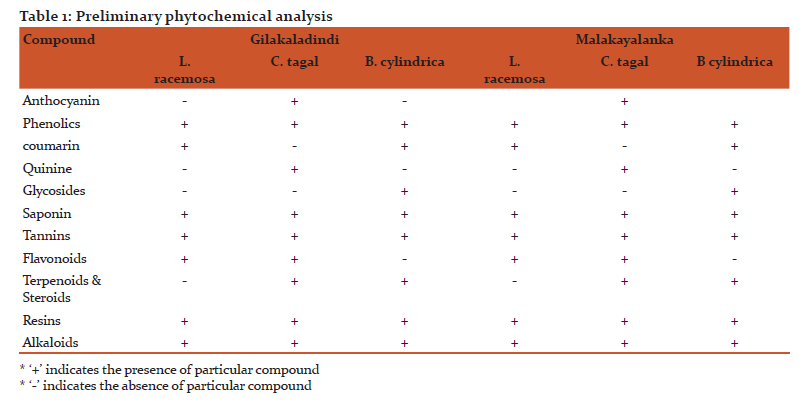
Antimicrobial activity
Antibacterial and antifungal activities of selected mangrove plants were studied and analyzed. The present study revealed the significant influence of seasons on potential antimicrobial activity of plant extracts. All the test organisms responded varied to different plant extracts at different concentrations of crude extracts i.e 50 μl, 100 μl, 150 μl and maximum inhibitory action was obtained at 150 μl.
Summer season
The leaf methanolic extracts of Gilakaladindi showed significant inhibiton activity. The plant extracts of mid-summer reported antimicrobial activity ranged from 7-11 mm with a mean average of 9.06 mm at 50 μl concentration. At 100 μl plant extract concentration the sensitivity of test organisms ranged from 8-13 mm with an average mean of 10.26 mm. On the other side at 150 μl the maximum inhibition was ranged from 8-15 mm with a mean average 11.00 mm. The extracts of C. tagal showed potential antimicrobial activity against E. coli (11.00±1.25 mm), S. aureus (13.00±1.00 mm) and B. cereus (15.00±0.25 mm) at 50 μl, 100 μl and 150 μl concentrations respectively (Figure 1). In case of Malakayalanka antimicrobial activity ranged between 7-10 mm with a mean average of 8.26 mm at 50 μl concentration. At 100 μl plant extract concentration microbial activity ranged from 7-12 mm with an average mean of 9.00 mm. At 150 μl the maximum zone of inhibition was ranged from 8-14 mm with a mean average 9.66 mm. At 50 μl concentration, leaf extract of C. tagal recorded maximum antimicrobial activity with S. aureus, E. coli and C. albican (10.00±1.102). Test organisms B. subtilis and E. coli showed highest sensitivity (12.00±0.958) when medium diffused with 100 μl leaf methanolic extracts of C. tagal. When all the test organism treated with 150 μl methanolic leaf extracts C. tagal extracts found to be more potential against B. subtilis with a zone of inhibition of 14.00±0.085 (Figure 2). The results of the present study indicated that the methanolic leaf extracts of summer season of Gilakaladindi showed more inhibiton activity when compared to Malakayalanka.
Rainy season
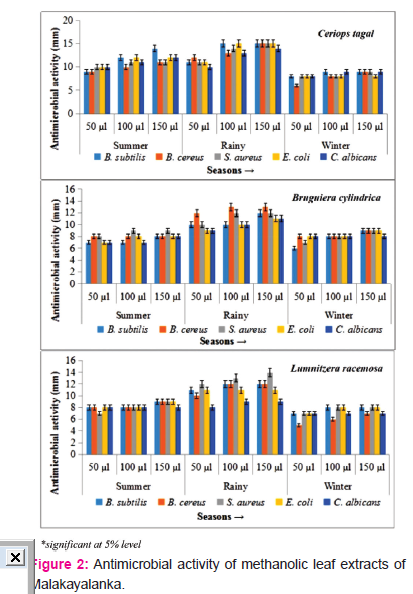
Rainy season showed highest positive antimicrobial activity and antifungal activity in all the plant species during all the seasons in both Gilkaladindi and Malakayalanka stations. The methanolic extracts of peak rainy season of Gilakaladindi, reported a varied antimicrobial activity 9-13 mm with a average mean of 11.13 mm at 50 μl concentration whereas at 100 μl concentration the inhibitory activity differed between 12-16 mm with an average mean of 14.13 mm. At 150 μl concentration of plant extract the maximum sensitivity is occurred between 14-18 mm with a mean average 15.6 mm. Samples collected during rainy season showed maximum inhibitory activity against all the test organisms. At 50 μl and 100 μl concentrations, leaf methanolic extracts of C. tagal showed high microbial activity 13±0.00 and 16±0.45 against B. subtilis and S. aureus respectively. Increased concentration of methanolic leaf extracts i.e 150 μl especially C. tagal extracts potentially increased the inhibitory activity (18.00±0.00) in B. subtilis and B. cereus (Figure 1) (Plate 1 A, B). Samples collected from Malakayalanka are not potentially bioactive when compared with Gilakaladindi samples in combating harmful microorganisms (Figure 2). In case of Malakayalanka antimicrobial activity varied from 8-12 mm with an average mean of 10.46 mm at 50 μl concentration. At 100 μl concentration inhibitory activity ranged from 9-15 mm with an average mean of 12.13 mm. The potential inhibitory activity at 150 μl concentration ranged from 9-15 mm with a mean average 12.8 mm. B. Cereus (12.00±0.12 mm) strains found to be more sensitive against C. tagal and B. cylindrica at 50 μl concentration. Methanolic leaf extract of C. tagal recorded maximum antimicrobial activity with B. subtilis and E. coli (15.00±0.04) at 100 μl concentration. In case of 150 μl concentration leaf extracts of C. tagal recorded maximum antibacterial activity (15.00±0.00) on S. aureus (Plate 1C), and against fungal organism C. albican (14.00±0.02) (Figure 2) (Plate 1D). Results revealed that the leaf methanolic extracts of rainy season are more sensitive against Bacillus subtilis and Bacillus cereus at Gilakadindi and S. aureus and C. albican at Malakayalanka stations.
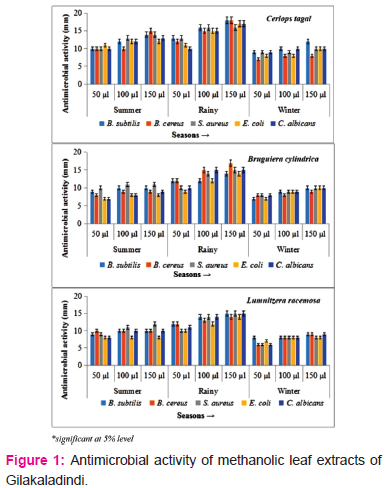
Winter season
In winter season all the extracts recorded lowest antibacterial and antifungal activity when compared to summer and rainy seasons. In case of Gilakaladindi antimicrobial activity differed between 6-9 mm with a mean average of 7.53 mm at 50 μl concentration. Methanolic leaf extracts 100 μl concentration recorded antimicrobial activity ranged from 8-10 mm with an average mean of 8.6 mm. At 150 μl concentration of leaf extract antimicrobial sensitivity was ranged from 8-12 mm with a mean average of 9.46 mm. Of all the plants highest microbial activity was reported with C. tagal leaf methanolic extracts in all three concentrations. At 50 μl maximum inhibitory activity (9.00±1.24 mm) was observed against B. subtilis, S.aureus, C. albicans whereas at 100 μl concentration strains B. subtilis, C. albicans recorded more inhibition (10.00±0.110). In case of 150 μl concentration, leaf extract of C. tagal recorded maximum antimicrobial activity with B. subtilis (12.00±0.00) (figure 1). Plant extracts collected from Malakayalanka showed relatively less activity when compared to Gilakaladindi. At 50 μl concentration the mean antimicrobial activity was ranged from 6-8 mm, with a mean of 7.20 mm. At 100 μl, 150 μl concentrations antimicrobial activity varied between 6-9 mm, 7-9 mm and with a mean average of 7.93 mm and 8.40 mm respectively. Except E. coli all the strains showed maximum sensitivity (9.00±0.11) towards C. tagal extracts when compared to other two plant extracts.
Nutrient analysis
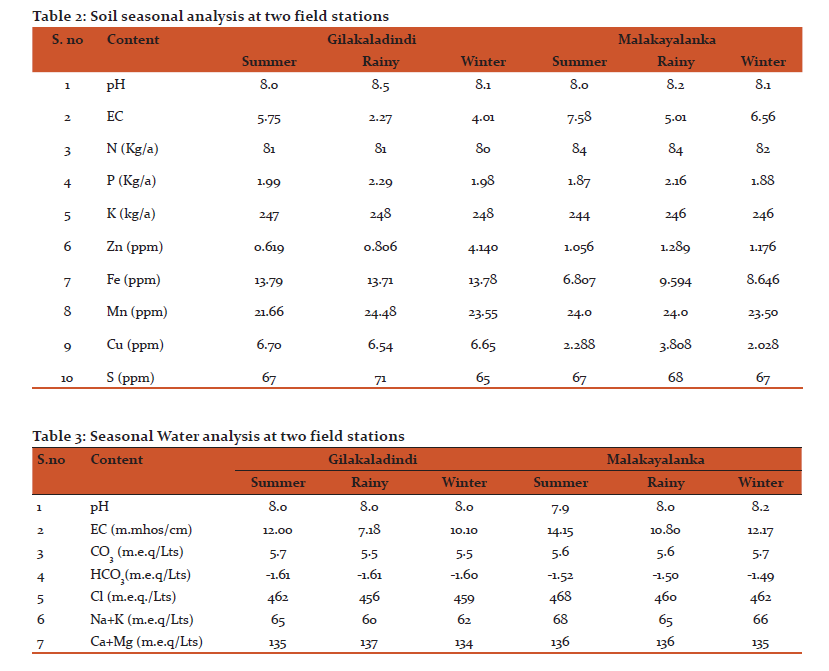
Both in Gilakaladindi and in Malakayalanka field stations the available soil and water nutrients were measured and tabulated. As compared with Malakayalanka the soil samples of Gilakaladindi contained less nitrogen (S:81 kg/a; R:81 kg/a and W:80 kg/a), more phosphorus (S:1.99 kg/a; R:1.99 kg/a and W:1.98 kg/a), zinc (S:1.015 ppm; R:1.019 ppm and W:1.015 ppm), iron (S:13.79 ppm; R:13.77 ppm and W:13.78 ppm) and copper (S:6.70 ppm; R:6.54 ppm and W:6.65 ppm). The availability of manganese (S:24.0 ppm; R:24.48 ppm and W:23.50 ppm) and sulphur (S:67 ppm; R:68 ppm and W:67 ppm) were found to be more in Gilakaladindi in all the seasons. The availability of all these components were increased during rainy season when compared to summer and winter seasons (Table 2 and Table 3). Electrical conductivity is an important parameter to evaluate the water quality. Any change in the concentration of chlorides and sulphates may influence the changes in the conductivity. In present study significant variation in electrical conductivity was observed in all the seasons (Table 2 and Table 3). The electrical conductivity values were found to be recorded more during summer in both Gilakaladindi (5.75 m.mmhos/cm) and Malakayalanka (7.58 m.mmhos/cm) stations. Among all the components the concentration of CO-3 was increased slightly (5.7 m.e.q/Lts) during winter season in Malakayalanka region. The availability of chlorides, sulphates, sodium, potassium were slightly decreased during rainy seasons both in Gilakaladindi and Malakayalanka regions (Table 2 and Table 3).
DISCUSSION
In mangrove ecosystem nutrients are considered as the significant components that influence growth and development of the plants.[23] and their availability is majorly based upon season, tidal forces and influx of fresh water. In present study also the availability of nutrients found to be increased during rainy season (Table 2). This increased nutrient availability during rainy season is may be due to influx of not only fresh water but also due to organic minerals carried by the rain water. Moreover the availability of nutrients observed to be more at Gilakaladindi as compared with Malakayalanka is because of high tidal currents at Gialakaladindi when compared to Malakayalanka. Frequent changes in water sources from ocean to fresh water or vice-versa may have significant effect on growth and production of bioactive compounds and result in seasonal variations in microbial activity.[24,25] Seasonal variations in protein, polyphenol and tannin concentration in leaves of Rhizophoraceae was reported and the organic constituents proteins, polyphenol and tannin concentration were generally highest in rainy seasons, intermediate in summer and lowest in winter of mangrove leaves.[26] Earlier studies indicated that anti bacterial activity of the leaves of Ceriops tagal showed 9.78±0.32 mm zone of inhibition against Bacillus cereus and 9.25 ±0.84 mm against Staphylococcus aureus[27,28]. Bruguiera cylindica showed zone of inhibition of 6.8 ±0.84mm against Pseudomonas aeruginosa.[29,30] Highest antibacterial activity of aqueous and methanolic extracts of leaves and shoots of Lumnitzera racemosa and Ceriopsis decandra was previously reported.[31,32] In the present study also rainy season samples showed highest overall mean zone of inhibition of 13.62 mm and 11.79 mm. The high concentration of organic constituents in leaf extracts could be the reason for pronounced microbial activity in rainy season. Excoecaria agallocha leaf extracts of rainy season collected from mumbai coast reported highest antimicrobial and anti fungal activity in Klebisella pneumonia, Aspergillus flavus and Phytopthora infestans.[33] In present study also irrespective of location, samples collected during rainy season showed more activity compared to summer and winter samples. In fact samples collected from Gilakaladindi station showed comparatively more antimicrobial activity over samples of Malakayalanka.
CONCLUSION
The present study concluded that the seasonal and geographical variations were found to be significant in production secondary metabolites. Of all the seasons rainy season is relatively more preferable for sample collection followed by summer. As the methanolic leaf extracts of rainy season showed highest inhibitory activity on bacterial and fungal growth at both sampling sites. Among the three mangrove species investigated leaf extracts of Ceriops tagal showed more inhibitory activity followed by Lumnitzera reacemosa and Bruguiera cylindrica. Further research in isolation and quantification of secondary metabolites from the mangrove leaf samples of rainy season would provide information in developing natural antibiotics with therapeutic potential and pharmacological efficacy.
ACKNOWLEDGEMENT
Author acknowledges the immense help received from the scholars whose articles are cited and included in references of this manuscript. The author is also grateful to authors / editors / publishers of all those articles, journals and books from where the literature for this article has been reviewed and discussed.
Conflict of Interest: Authors have no conflict of interest
References:
-
John MM, Karin RB, Erik MH and Julia CM. Attenuation of tides and surges by mangroves: Contrasting case studies from New Zealand. Water 2018; 10, 1119. doi:10.3390/w10091119.
-
Naidoo G, Rogalla H and Von Willert DJ. Gas exchange responses of a mangrove species, Avicennia marina, to waterlogged and drained conditions. Hydrobiologia 1997; 352(1-3): 39-47.
-
Seedo KA, Mohammad SA, Ahmed S and Asma A. Morphophysiological traits of gray mangrove (Avicennia marina (Forsk.) Vierh.) at different levels of soil salinity. Intl J Forest Res 2018; 1-9. https://doi.org/10.1155/2018/7404907.
-
Lima YB, Francisco VSS, Miguel FN, Emanoela PP and Hans RG. Accumulation of salts in the soil and growth of cowpea under salinity and phosphorus fertilization. Revista Ciencia Agronomica 2017; 48(5): 65-773.
-
Haque MA, Mezanur MR, Nihad SAI, Howlader MRA., Akand MMH. Morpho-physiological response of Acacia auriculiformis as influenced by seawater induced salinity stress. Forest System 2016; 25(3):71-79.. http://dx.doi.org/10.5424/fs/2016253-09386.
-
Basak U, Das AB, Premananda D. Chlorophylls, carotenoids, proteins and secondary metabolites in leaves of 14 Species of mangrove. Bull Marine Science 1996; 58(3):654-659.
-
Bryant JP, Chapin FSI, Klein DR. Carbon/nutrient balance of boreal plants in relation to vertebrate herbivory. Oikos 1983; 40:357-68.
-
Speed MP, Fenton A, Jones MG, Ruxton GD and Brockhurst MA. Coevolution can explain defensive secondary metabolite diversity in plants. New Phytologist 2015; 208:1251–1263. doi: 10.1111/nph.13560.
-
Lopez GX, Villari C, Bonello P, Borg AK, Grivet D, Zas R and Sampedro L. Inducibility of plant secondary metabolites in the stem predicts genetic variation in resistance against a key insect herbivore in maritime pine. Front Plant Science 2018; 9:1651. doi: 10.3389/fpls.2018.01651.
-
Bandaranayake W. Traditional and medicinal uses of mangroves. Mangroves Salt Marshes 1998; 2(3):133-148. https://doi.org/10.1023/A:1009988607044.
-
Abeysinghe PD. Antibacterial activity of some medicinal mangroves against antibiotic resistant pathogenic bacteria. Indian J Pharma Science 2010; 72(2): 167–172. doi: 10.4103/0250-474X.65019.
-
Ho DY, Vodouhe FG and Sinsin B. Ethnobotanical survey of mangrove plant species used as medicine from ouidah to grand-popo districts, Southern Benin. American J Ethnomedicine 2017; 4(1): 8,1-6.
-
Salini G. Pharmacological profile of mangrove endophytes - A Review. Intl J Pharma Pharmaceutical Science 2015; 7(1): 6-15.
-
Jayakumar, Malarvannan S, Suresh VM and Balasooriya NWB. 46th International Symposium 2016; South Eastern University of Sri Lanka, pp.738-748.
-
Reshma NK and Mani Murali R. Current status and decadal growth analysis of Krishna - Godavari delta regions using remote sensing. J Coastal Res. Special Issue No. 85: Proceedings of the 15th International Coastal Symposium, Haeundae, Busan, 13-18 May 2018; pp. 1416-1420 (5 pages). Published by: Coastal Education & Research Foundation Inc.. doi:https://www.jstor.org/stable/26488451.
-
Norman C. Duke NC, Ball MC, Ellison JC. Factors influencing biodiversity and distributional gradients in mangroves. Global Ecology Biogeography Lett 1998; 7(1):27-47. doi:www.jstor.org/stable/2997695.
-
Dangremond EM, Feller IC, Sousa WP. Environmental tolerances of rare and common mangroves along light and salinity gradients. Oecologia 2015; 1-12. doi: 10.1007/s00442-015-3408-1.
-
Vijaya Kumar K, Rosaiah G, Babu K, Tirupati Swamy N, Krishna Naragani. A Study on antimicrobial properties of herbal nano particles of selected mangrove plants. Res. J Life Science, Bioinformatic, Pharmaceutical and Chemical Science 2018; 4(5):498-512.
-
Harborne JB. Phytochemical Methods, London, Chapman and Hall Ltd.,1973; 49-188.
-
Gibbs RD. Chemotaxonomy of flowering plants, McGill queen’s university press, Montreal 1974; 523-619.
-
Hunter. New techniques and equipment for routine soil plant analytical procedures. Elemer Bornemisza and Alfredo Alvarado. Soil manage Tropical America 1975; Proceedings of a Seminar held at CIAT, Cali, Colombia February 10 - 14, 1974, 467-483.
-
Motsara MR, Roy RN. Guide to laboratory establishment for plant nutrient analysis. FAO Fertilizer Plant Nutrition Bull, Food Agrl Org 2008; Rome.
-
Arumugam A and Sugirtha PK. Evaluation of physico-chemical parameters and nutrients in the mangrove ecosystem of Manakudy Estuary, South West coast of India. Intl J Latest Res Science Tech 2014; 3 (6):205- 209.
-
Kirchman DL, Malmstrom RR, Cottrell RR. Control of bacterial growth by temperature and organic matter in the Western Arctic. Deep Sea Research Part II Topical Studies Ocean 2005; 52(24):3386-3395. doi: 10.1016/j.dsr2.2005.09.005.
-
El-Fatimy ES, Said AA, and Massoud MG. Seasonal variation and antifungal activities of methanolic algal extracts of some dictyotaceae of Benghazi coasts, Libya. Egyptian J Phycol 2009;10(1-9).
-
Basak UC, Das AB, Das P. Seasonal changes in organic constituents in leaves of nine mangrove species. Marine Freshwater Res 1998; 49(5):369-372.). doi:10.1071/mf97029.
-
Ramteke L, Jadhav BL and Poonam G. Biogenic copper nanoparticles from the aqueous stem extract of Ceriops tagal. World J Pharma Res 2018; 7(18): 933-947.
-
Sumardi, Mohammad B, Ridha W. Antimicrobial activity of polyisoprenoids of sixteen mangrove species from North Sumatra, Indonesia. Biodiversitas 2018; 19(4):1243-1248.
-
Millat MS, Islam S, Hussain MS, Moghal MMR, Islam T. Anti-bacterial profiling of Launaea sarmentosa (Willd.) and Bruguiera cylindrica (L.): Two distinct Ethno medicinal plants of Bangladesh. European J Exptll Bio 2017; 7(1):1-5.
-
Satarupa R, Madhumita R, Prosenjit P, Bulti N and Abhijit M. Antimicrobial activity and phytochemical constituents of Bruguiera gymnorhiza fruit collected from Indian Sundarbans, the designated World Heritage site. Intl J Green Herbal Chem 2018; Sec. B; 7(2):119-125. doi: 10.24214/IJGHC/HC/7/2/11925.
-
Anil K, Ammani K, Siddhardha B. In vitro antimicrobial activity of leaf extracts of certain mangrove plants collected from Godavari estuarine of Konaseema delta, India Vundru. Intl J Med Aromatic Plant 201; 1(2):132-136.
-
Jasna TK, Khaleel KM and Rajina M. In vitro antibacterial activity of mangrove plant Kandelia candel (L.) druce (Rhizophoraceae). World J Pharma Res 2017; 6(11):470-477.
-
Yeragi L.A and Mendhulkar VD. Seasonal variations in antibacterial and antifungal activity of Excoecaria agallocha. Asian J Microb, Biotech, Environ Science 2017; 19(1):111-115.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License