IJCRR - 2nd Wave of COVID-19: Role of Social Awareness, Health and Technology Sector, June, 2021
Pages: 182-187
Date of Publication: 11-Jun-2021
Print Article
Download XML Download PDF
Evaluation of the Effectiveness of Rapid Diagnostic Test Antibody, Serological Test, Reverse-Transcribed Polymerase Chain Reaction (RT-PCR) and Hematology in Non-Severe Covid-19 Patients
Author: Elisabeth LS Setianingrum, Kartini Lidia, Kristian Ratu
Category: Healthcare
Abstract:Introduction: COVID-19 is a zoonotic disease caused by a coronavirus which is similar to Middle East Respiratory Syndrome (MERS) and Severe Acute Respiratory Syndrome (SARS), which can spread mainly from person to person through droplets from the nose or mouth through coughing, sneezing or talking and inhaling droplets from an infected person. Antibody Rapid Diagnostic Test (RDT) is a simple, inexpensive and efficient screening kit. It employs whole blood or serum/blood plasma samples as a humoral immunological response. If the result is reactive then it will be followed by a Reverse Transcriptase-Polymerase Chain Reaction (RT-PCR) examination. Evaluation of the haematological profile of COVID-19 patients is also important for patients with mild or no symptoms. Aims: To evaluate and compare the effectiveness of RDT Antibody, serological testing, RT-PCR and haematology in non-severe covid-19 patients. Methodology: The study design was a cross-sectional study that was conducted from October to December 2020 in several hospitals in the city of Kupang which treated patients with mild or no symptoms. Research respondents were 34 subjects who had agreed and signed the informed consent. The subjects were taken their blood samples for RDT antibody examination using blood serum (taken from centrifuged whole blood and separated the blood serum), where the reactive results on RDT antibodies must be followed by confirmatory or diagnostic tests which are the nasopharyngeal and oropharyngeal swabs for reverse�transcribed Polymerase Chain Reaction(RT-PCR)examination. Furthermore, for the LED examination which is the Complete Hematology, 3 ml of whole blood was taken and then processed. Results: The comparison between RDT antibody and SARS CoV2 serology was 73.5%. While RDT and the RT-PCR results of 47.1%; and RT-PCR with serology was 70.6%. Conclusion: RDT antibody and serology SARS COV2 detect the presence of antibodies that have been formed by the body approximately 1-2 weeks after the acute infection. RT-PCR is the best diagnostic to detect the presence of the virus in the respira�tory tract during acute infection and is followed by the patient's clinical condition. Haematological analysis shows the prognostic and severity of COVID19 patients. Patients with mild symptoms / no symptoms do not have much difference in haematology results than normal people.
Keywords: Rapid diagnostic test, Antibody, RT-PCR, Serology, Hematology
Full Text:
INTRODUCTION
COVID-19 is a zoonotic disease caused by a coronavirus which is similar to Middle East Respiratory Syndrome (MERS) and Severe Acute Respiratory Syndrome (SARS), which can spread mainly from person to person through droplets from the nose or mouth through coughing, sneezing or talking and inhaling droplets from an infected person. COVID-19 originated from an outbreak in Wuhan, China, in December 2019 and continues to spread throughout the world, to Indonesia on March 2, 2020, and on April 9 2020 patient zero was recorded in Kupang City, East Nusa Tenggara. Until now, all hospital isolation rooms are filled with COVID-19 patients. 1,2,3
Clinical manifestations of COVID-19 vary widely, starting from the incubation period of approximately 3-14 days until the onset of symptoms, such as mild cough, runny nose, fever, anosmia, ageusia, or even no symptoms at all to severe respiratory symptoms such as acute respiratory distress syndrome (ARDS). From March to the end of December 2020 Kupang City started treating people with mild and severe symptoms. Based on the Guidelines for Handling COVID-19 of the Indonesian Ministry of Health 4th Edition, a screening tool such as the Rapid Diagnostic Test (RDT) antibody is used. If the result is reactive then it will be followed by a Reverse Transcriptase-Polymerase Chain Reaction (RT-PCR) examination. Antibody Rapid Diagnostic Test (RDT) is a simple, inexpensive and efficient screening kit. It employs whole blood or serum/blood plasma samples as a humoral immunological response. There are lots of commercial RDTs circulating the market with different brands, prices, sensitivities and specificities.1-5
The presence of antibodies as the body's humoral response to COVID19 infection shows whether there has been a recovery in the patient, besides that it can be used as a screening for asymptomatic conditions such as a study conducted by Guo et al. by detecting the emergence of Immunoglobulin (Ig) A, M, G. Another study by Tofe et al. on commercial RDT was tested on sensitivity and specificity by comparing RT-PCR examinations. The study by Elslande et al. compared seven commercial RDT IgM / IgG antibodies against the IgA / IgG Elisa tool to obtain the sensitivity and specificity of each RDT.5-8
Evaluation of the haematological profile of COVID-19 patients is also important for patients with mild or no symptoms. Haematological parameters such as haemoglobin, leukocytes, platelets, sedimentation rate (ESR), neutrophils, lymphocytes, monocytes and neutrophil lymphocytes ratio (NLR), platelet lymphocyte ratio (PLR) are used as biomarkers and prognostic factors to assess the severity of the disease in Covid-19 patients.9-12
This study will evaluate the use of several commercial RDT antibodies and compare them with the emerging serologic results examined using the Electro-Chemiluminescence Immunoassay (ECLIA) method and the suitability of the RT-PCR results in mild symptomatic and asymptomatic patients admitted to several Kupang city hospitals at the end of 2020 and the emerging haematological profile.
MATERIALS AND METHOD
The study design was a cross-sectional study that was conducted from October to December 2020 in several hospitals in the city of Kupang which treated patients with mild or no symptoms. The study was approved by the Ethics Commission of the Faculty of Medicine of the University of Nusa Cendana 2020. Research respondents were 34 subjects who had agreed and signed the informed consent.
The subjects were taken their blood samples for RDT antibody examination using blood serum (taken from centrifuged whole blood and separated the blood serum), where the reactive results on RDT antibodies must be followed by confirmatory or diagnostic tests which are the nasopharyngeal and oropharyngeal swabs for RT-PCR examination. Furthermore, for the LED examination which is the Complete Hematology, 3 ml of whole blood was taken and then processed.
The RDT antibody examination used the lateral flow method which analyzes the suitability of several commercial RDTs used in various health services with reactive results when antibodies are present and non-reactive if antibodies have not been formed. Subsequently, RT-PCR examination is carried out to check for any viruses in the patient's respiratory tract with a positive result if the virus is present (a patient confirmed with COVID-19) and negative if there is no virus.
Serological levels of SARS CoV2 were examined using the anti-SARS COV2 Electro-Chemiluminescence Immunoassay (ECLIA) method to detect antibodies in serum as an adaptive immune response to the SARS COV2 virus. This examination is a semiquantitative examination by comparing the electrochemiluminescence signal of the antigen-antibody reaction in a sample with a cut off index (COI) limit value if COI ≥ 1 indicates a reactive/positive state if COI <1 indicates a non-reactive / negative state, which is adjusted for clinical condition and physical examination of the patient.13 The RT-PCR used in the examination used the closed system method with 96 well examinations with the automatic Abbott m2000 system, which minimizes contamination and reduces the incidence of false positives, besides that the RT-PCR results also require clinical information and a physical examination that supports the results of the examination.14
RESULTS AND DISCUSSION
As shown in Table 1, this study comprised of 34 subjects which consisted of various age categories, most of which were dominated by the age of 21-30 years old by 35.3%; with the youngest age at 19 years and the oldest at 63 years old. This is because this age is the productive age thus many are exposed to other people. A total of 70.6% were examined for haematological profile and RT-PCR, while the others did not because the patients were asymptomatic and in good clinical condition, with oxygen saturation of 97-98%.
The results of the RT-PCR, namely the examination of the Nucleic Acid Amplification Test (NAAT) by detecting the presence of the virus, obtained 26 positive confirmed COVID-19 results (76.5%) with a Cycle Threshold (CT) value below 31.5.15,16,17
The emergence of antibodies that can be seen from serological examination with the ECLIA method, the reactive results are more with serological levels of COI> 1, while respondents with serological levels of COI <1 were only 14 people (41.2%). This is due to an indication of patient care in the hospital, namely RDT reactive antibodies, in patients who have COVID-19 infection the appearance of antibodies as a body response occurs a few days to weeks after infection, thus the serological level of SARS COV2 is also high>1, but the appearance of antibodies in the body depending on nutritional factors, patient age, the severity of disease and treatment are given to patients. SARS COV2 antibody and serological RDT tests cannot be used to make a clinical diagnosis, because the acute infection period has passed, often causing false-positive results due to cross-reactions with other types of pathogens that cause coronavirus.15,18
RDT antibodies with the lateral flow method vary in the results released due to the differences in the quality of nitrocellulose and recombinant proteins and their stability when reagent stored. Besides that,false-negative results can occur in patients with severe symptoms. On the contrary, false-positive results can occur in cross-reactions to other types of coronavirus pathogens. Great suitability indicates that the selection of RDT antibodies used by the hospital in this study is quite good even though only a few reagents are used, however, the RDT antibody test is not recommended by WHO for clinical diagnosis because it shows more acute past infections.5,15,16,19

The comparison between RDT antibody and SARS CoV2 serology was 73.5%. This is because they both detect the presence of antibodies in the blood, where the antibodies that appear are IgM and IgG. IgM appears a few days after infection and is followed by the IgG, while serology tests can see the levels of antibodies that have been formed.5-7,16,20
Table 2 depicts the conformity between SARS CoV2 and RT-PCR serology was 70.6%, while the smallest conformity occurred between the use of RDT and the RT-PCR results of 47.1%. This is due to the differences in the examination samples used and different examination methods. The relatively high results between serology and RT-PCR results indicate that these two methods are acceptable to be used to see the emergence of antibodies and the acute phase that occurs while the virus is still present, meanwhile RDT results are less reliable in detecting the appearance of antibodies in a person's body.6,8,15,17
Antibodies are formed after the body forms immunity against viruses that enter the body. Due to the limitation of diagnostic tools such as rapid tests, it is important to continue the diagnosis with the RT-PCR examination (molecular test) to determine precisely the presence of the virus and the ability to replicate through the CT value of the SARS COV2 virus. According to WHO, molecular tests are the most recommended tests to diagnose COVID-19. Therefore, screened patients using the antibody Rapid test with reactive results should be carried on with RT-PCR examination. Those with a non-reactive rapid test result means that antibodies have not been formed now or in the past.6,18
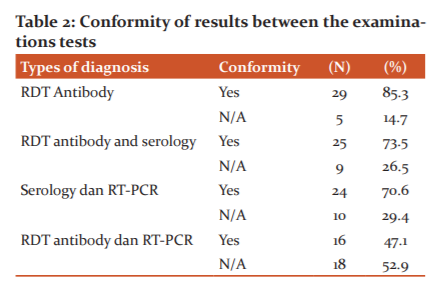
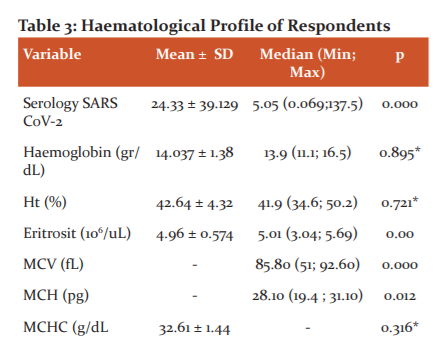
Table 3 shows that the serological levels of SARS COV2 in the subjects had a mean value of 24.33 ± 39.129 where some of the patients had high serological levels of SARS COV2 (COI> 1), this shows the body's good immunological response to viruses in the body. Low antibody levels (COI <1) indicated that antibodies have not been formed. The highest antibody level was at COI 186.4, while the lowest COI antibody level was 0.062 owned by subjects of acute phase COVID-19 (antibodies had not been formed). Other respondents on the first day of the acute phase of treatment had positive RT-PCR results with low serological levels of 0.086 but compared to the last day of treatment after four weeks of independent isolation, these respondents obtained negative RT-PCR results and high serological antibody levels, which is 29.550. Another respondent without symptoms but had close contact with a COVID19patient had a negative RT-PCR but the serologic result was high, which is COI 106.7, indicating the formation of antibodies was quite high from the first close contact with a COVID-19 patient in the past. Haematological evaluation of patients with confirmed COVID-19 with RT-PCR were 24 people with the results in table 3. In the haematological description of the study, respondents showed that the results with haemoglobin levels had an average of 14.037 ± 1.38 with a morphological picture of erythrocytes with an average MCV, MCH, MCHC is normal (normochromic normocytic), although there is one subject with moderate anaemia due to a previous chronic disease and microcytic and hypochromic erythrocyte appearance. The above-normal hematologic conditions do not occur in patients with severe COVID19 symptoms, where those with severe symptoms experience anaemia, due to iron dysmetabolism, a state of ferroptosis and the influence of a state of oxidative stress due to viral influences.21-22
The subjects’ leukocytes have a normal range with normal data distribution without any extreme values. The leukocyte count (eosinophils, basophils, neutrophils, lymphocytes, monocytes) also are within the normal range due to the mild symptoms or without symptoms in the subjects. One subject had a lower lymphocyte count of 2.49% compared to the other subjects. Lymphopenia can occur in COVID-19 patients due to a decrease in lymphocyte effectiveness. Lymphopenia occurs in 85% of cases of severe COVID-19 infections due to a decrease in the number of Natural Killer (NK) cells and T lymphocytes so that the natural and adaptive immunological response to kill the virus decreases. The number of lymphocytes decreases with the severity of the disease due to severe cytokine storms, thus COVID-19 patients with mild symptoms have more lymphocytes than severe symptoms as in this study population.11-12
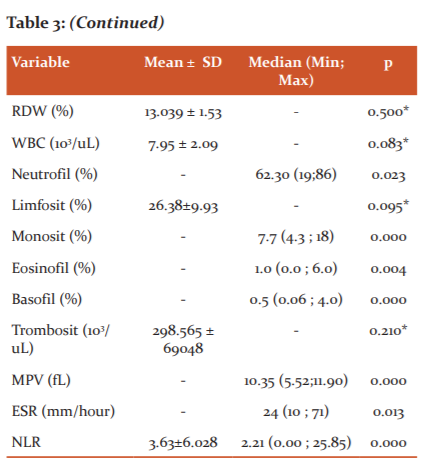
In contrast to this study, the haematological conditions that occur in severe COVID-19 infections are leukocytosis with neutrophilia which is the cause of cytokine storms and hyperinflammatory conditions that aggravate the condition of patients with severe COVID-19 symptoms. Besides morphological disorders such as hypopigmentation in neutrophilia which indicate a bacterial infection. Neutrophils, which are the largest part of leukocytes, if there is an increase, will induce DNA cell damage due to the release of Reactive Oxygen Species (ROS) and trigger the release of inflammatory factors such as Interleukins (IL-6, IL-8, TNF, Inferon Gamma) from lymphocytes and endothelial cells. This increased neutrophil and decreased lymphocyte count resulted in an increased neutrophil-lymphocyte ratio (NLR).10
The state of inflammation or bacterial infection can be seen from the Neutrophil Lymphocyte Ratio (NLR), wherein in severe conditions the NLR value is> 3.13. In this study, the average NLR of all subjects was 3.33 ± 5.23. This suggests an inflammatory state that has also occurred in the treated patient. One respondent experienced a high NLR increase up to 25.85 due to a state of neutrophilia and lymphocytopenia. According to Kazancioglu et al, (2021) in their study, the lymphocyte, NLR and PLR values ??provide more clinical value than other parameters in differentiating patients with COVID-19 from another influenza, whereas according to Ghahramani et al, (2020) NLR determines prognostic condition in a disease.2,9,10,12
In this study, an increased mean number indicates that inflammation has occurred, although many factors can influence this increase in ESR results. This examination has often been replaced by other inflammatory markers such as CRP, PCT, LDH and NLR which also have an increase in inflammatory processes / other acute bacterial infections.9,11,12,23 The platelet count in this study population had a normal mean number, in contrast to COVID-19 patients with severe symptoms of thrombocytopenia, as occurs in other severe infectious diseases. Increased Platelet Lymphocyte Ratio (PLR) is also more indicative of disease severity than NLR.9
CONCLUSIONS
RDT antibody and serology SARS COV2 detect the presence of antibodies that have been formed by the body approximately 1-2 weeks after the acute infection. RT-PCR is the best diagnostic to detect the presence of the virus in the respiratory tract during acute infection and is followed by the patient's clinical condition. Haematological analysis shows the prognostic and severity of COVID19 patients. Patients with mild symptoms / no symptoms do not have much difference in haematology results than normal people.
ACKNOWLEDGEMENTS
This research was fully funded and supported by the University of Nusa Cendana, Indonesia.
CONFLICT OF INTERESTS
There is no conflict of interests found during this study
ETHICS
This study has received ethical approval from the Health Research Ethics Commission of the Faculty of Medicine, University of Nusa Cendana.
AUTHOR CONTRIBUTIONS
Elisabeth was responsible in the whole project, including the protocol. Kartini and Kristian contributed in data analysis and writing. Elisabeth supervised the experiment and wrote the whole manuscript.
References:
1. Ministry of Health, Republic of Indonesia. Guidelines for the Prevention and Control of Corona Virus Diseases (Covid-19). 2020;5:178. Available from: https://covid19.go.id/storage/app/media/Protokol/REV-05_Pedoman_P2_COVID-19_13_Juli_2020.pdf
2. Susilo A, Rumende CM, Pitoyo CW, Santoso WD, Yulianti M, Herikurniawan H, et al. Coronavirus Disease 2019: Recent Literature Review. J Peny Dal Ind. 2020;7(1):45.
3. Bere SM. First Positive Covid-19 Patient in NTT, This YouTuber Announces Swab Test Results on YouTube. Kompas.com [Internet]. 2020; Available from: https://kupang.kompas.com/read/2020/04/10/09221081/jadi-pasien-positif-covid-19-pertama-di-ntt-youtuber-ini-umumkan-hasil-tes?page=all.
4. Ministry of Health, Republic of Indonesia. Covid REV-4 Guidelines. Guidelines for the Prevention and Control of Coronavirus Dis. 2020; 1 (4th Revision): 1–125.
5. Pavlova IP, Nair SS, Kyprianou N, Tewari AK. The Rapid Coronavirus Antibody Test: Can We Improve Accuracy? Front Med. 2020;7(September):1–5.
6. Guo L, Ren L, Yang S, Xiao M, Chang D, Yang F, et al. Profiling early humoral response to diagnose novel coronavirus disease (COVID-19). J Clin Infect Dis. 2020;71(15):778–85.
7. Hoffman T, Nissen K, Krambrich J, Rönnberg B, Akaberi D, Esmaeilzadeh M, et al. Evaluation of a COVID-19 IgM and IgG rapid test; an efficient tool for assessment of past exposure to SARS-CoV-2. Infect Ecol Epidemiol [Internet]. 2020;10(1). Available from: https://doi.org/10.1080/20008686.2020.1754538
8. Elslande J Van, Houben E, Depypere M, Brackenier A, Desmet S, Andr E, et al. Diagnostic performance of seven rapid IgG / IgM antibody tests and the Euroimmun IgA / IgG ELISA in COVID-19 patients. 2020;(January).
9. Kazancioglu S, Bastug A, Ozbay BO, Kemirtlek N, Bodur H. The Role of Haematological Parameters in Patients with COVID-19 and Influenza Virus Infection. Epidemiol Infect. 2021;148(e272):1–8.
10. Yang A, Liu J, Tao W, Li H. The diagnostic and predictive role of NLR, d-NLR and PLR in COVID-19 patients. Int Immunopharmacol 84 [Internet]. 2020;84(January):1–7. Available from: https://doi.org/10.1016/j.intimp.2020.106504
11. Fathi N, Rezaei N. Lymphopenia in COVID-19: Therapeutic opportunities. Cell Biol Int[Internet].2020;44(9):1792–1797.
12. Ghahramani S, Tabrizi R, Lankarani KB, Kashani SMA, Rezaei S, Zeidi N, et al. Laboratory features of severe vs. non-severe COVID-19 patients in Asian populations: A systematic review and meta-analysis. Eur J Med Res [Internet]. 2020;25(1):1–10. Available from: https://doi.org/10.1186/s40001-020-00432-3
13. Bolton JS, Chaudhury S, Dutta S, Gregory S, Locke E, Pierson T, et al. Comparison of ELISA with electro-chemiluminescence technology for the qualitative and quantitative assessment of serological responses to vaccination. Malar J [Internet]. 2020;19(1):1–13. Available from: https://doi.org/10.1186/s12936-020-03225-5
14. Abbott Diagnostics. Abbott RealTime SARS-CoV-2: Instructions for Use. US Food Drug Adm website [Internet]. 2020;(Ic):1–12. Available from: https://www.fda.gov/media/136258/download
15. Yanti B, Ismida FD, Sarah KES. Differences in antigen, antibody, RT-PCR diagnostic tests and molecular rapid tests for Coronavirus Disease 2019. J Kedokt Syiah Kuala. 2020;20(3):172–1777.
16. Carter LJ, Garner L V., Smoot JW, Li Y, Zhou Q, Salveson CJ, et al. Assay Techniques and Test Development for COVID-19 Diagnosis. ACS Cent Sci. 2020;6(5):591–605.
17. Tang Y, Schmitz JE, Persing DH, Stratton CW. Laboratory Diagnosis of COVID-19: Current Issues and Challenges. J Clin Microbiol. 2020;58(6):1–9.
18. WHO.Recommended Use of Immunodiagnostic Tests at Health Care Facilities (Point of Care) for COVID-19. April 8th [Internet]. 2020; (Scientific Statement): 1–4. Available from: https://www.who.int/docs/default-source/searo/indonesia/covid19/saran-penggunaan-tes-imunodiagnostik-di-fasyankes-(point-of-care)-untuk-covid-19.pdf?sfvrsn=a428857b_2
19. Li Z, Yi Y, Luo X, Xiong N, Liu Y, Li S, et al. Development and clinical application of a rapid IgM-IgG combined antibody test for SARS-CoV-2 infection diagnosis. J Med Virol [Internet]. 2020;92(9):1518–24. Available from: http://dx.doi.org/10.1002/jmv.25727
20. Jacofsky D, Jacofsky E, Jacofsky M. Understanding Antibody Testing for COVID-19. One Heal. 2020;(January).
21. Lippi G, Mattiuzzi C. Hemoglobin value may be decreased in patients with severe coronavirus disease 2019. Hematol Transfus Cell Ther [Internet]. 2020;42(2):116–7. Available from: https://doi.org/10.1016/j.htct.2020.03.001
22. Cavezzi A, Troiani E, Corrao S. COVID-19: haemoglobin, iron, and hypoxia beyond inflammation. A narrative review. Clin Pract. 2020;10(2).
23. Terpos E, Ntanasis-Stathopoulos I, Elalamy I, Kastritis E, Sergentanis TN, Politou M, et al. Hematological findings and complications of COVID-19. Am J Hematol. 2020;95(7):834–847.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License