IJCRR - 6(1), January, 2014
Pages: 89-94
Print Article
Download XML Download PDF
STUDYING ANTI RETRO VIRAL THERAPY A CAUSATIVE FACTOR FOR OSTEONECROSIS IN HIV INFECTED PATIENTS
Author: Vetrivel Chezian Sengodan
Category: Healthcare
Abstract:Background and Objectives: Avascular necrosis of femoral head is a common entity. Avascular necrosis of femoral head due to anti-retroviral therapy is an emerging health problem in the world. In India, avascular necrosis of femoral head due to anti-retroviral therapy is not yet reported. We report a case of bilateral avascular necrosis of femoral head as a complication of anti-retroviral therapy. Material and methods: A 41 year old female, a known case of pulmonary tuberculosis had anti tuberculous treatment. While she was on anti tuberculous treatment, she was diagnosed for human immune deficiency infection and put on stavudine, lamivudine and efavirenz [transcriptase inhibitors] and continued for one year. Results: In the follow up period of our patient, she was found to have low back pain and pain in right hip and limp and she was diagnosed of having bilateral avascular necrosis of the femoral head. The diagnosis was confirmed by biopsy of both the femoral heads which revealed avascular necrosis. Interpretation and conclusion: Till date avascular necrosis of the bone due to anti-retroviral therapy is not yet reported from India. We report this case for its rarity and to stress the role of MRI in early detection of avascular necrosis of hip in patients with anti-retro viral therapy.
Keywords: Anti-retroviral therapy, avascular necrosis, Nucleoside and Non-Nucleoside reverse transcriptase inhibitors.
Full Text:
INTRODUCTION
Avascular necrosis is termed as “osteonecrosis” indicates ischemic death of the bone as a result of insufficient arterial blood supply.[1] Risk factors associated with avascular necrosis have included corticosteroid use, alcoholism, intravenous drug use, smoking, antiphospholipid antibodies, hyperlipidemia, sickle cell anemia, radiation exposure and systemic lupus erythematosus. Metabolic factors such as hyperlipidaemia have also been strongly associated with use of protease inhibitors, nucleoside and non nucleoside reverse transcriptase inhibitors. Regardless of the aetiology avascular necrosis has become an emerging manifestation within the Human immuno deficiency virus population. [1, 2]
MATERIAL AND METHODS
A forty one year old female, presented with history of pain over the lower back for three months and pain in the right hip for one month. She had pulmonary tuberculosis three years back, and was treated with antituberculous treatment under category one of Revised National Tuberculous Control Programme. While she was on anti tuberculous treatment, patient was diagnosed to have Human immuno deficiency virus Infection and was started on antiretroviral drugs stavudine, lamivudine and efavirenz. At one year follow up after anti retroviral therapy she presented with pain in the lower back and right hip. There was no history of steroid intake, cardiac disease and radiation exposure. On clinical examination patient was found to have tenderness over the lower lumbar spine. There was no obvious deformity or swelling. There was no distal neurological deficit. On examination of both the lower limbs, tenderness was present over the scarpa’s triangle on right side with no fixed deformity. Extremes of movements were painful and restricted on the right hip joint. There was no limb length discrepancy. The left hip was clinically normal. Antalgic gait was noted. Laboratory parameters showed that there is no raise in acute phase reactants. CRP and ESR were normal. Complete blood count was within normal limits. CD4 count was 590 cells/mm3 . Lipid profile showed high triglyceride level (420mg/dl) and high cholesterol level (364mg/dl). Peripheral smear was also normal.
RESULTS
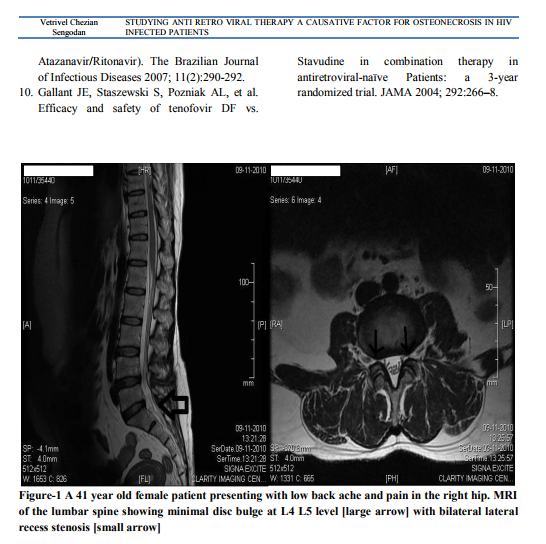
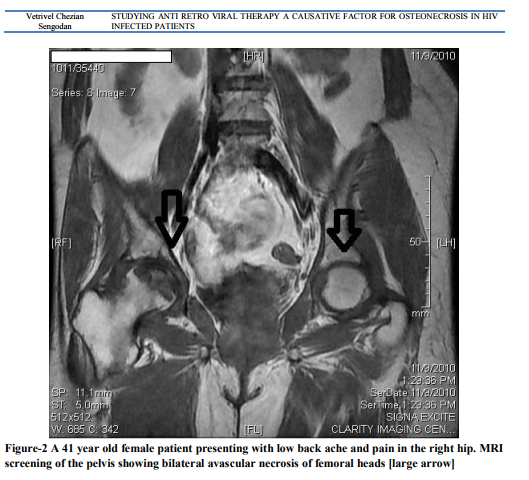
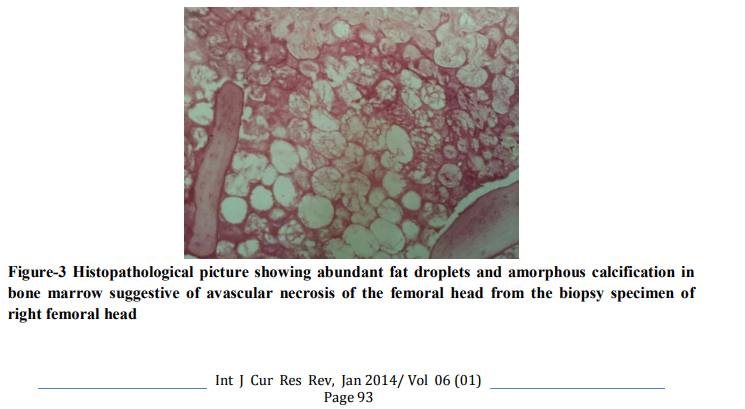
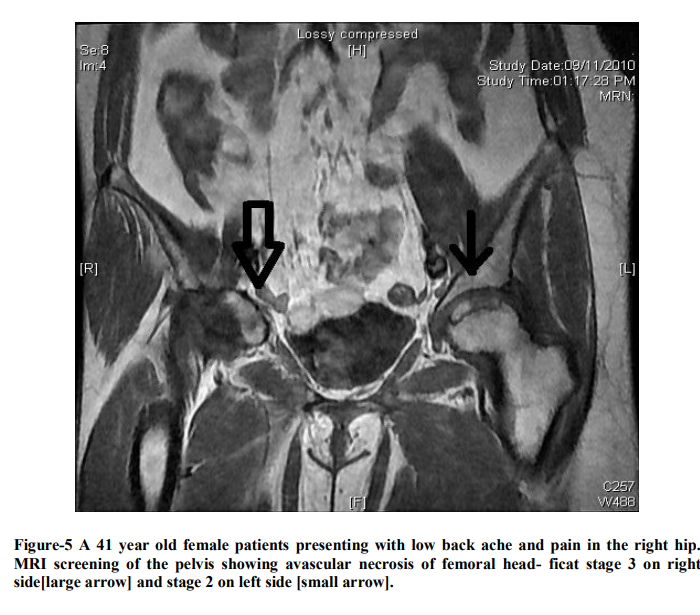
Radiograph of the pelvis showed increased density on the right hip joint, slight flattening of the weight bearing zone of the femoral head more on right side. MRI of the lumbar spine and screening of the pelvis showed minimal disc bulge at L4 L5 level with bilateral lateral recess stenosis[figure 1] and avascular necrosis involving both femoral heads[figure 2] which was confirmed by biopsy [figure 3 and 4]. As per Ficat and Arlet classification, it was stage 3 on right and stage 2 on left side [figure 5].
DISCUSSION
Anti retroviral therapy is the only treatment option available for human immuno deficiency infected individuals. Normally combinations of anti retroviral drugs are used. In our country, nucleoside and non nucleoside reverse transcriptase inhibitors are used as a first line of drugs for the diseased individuals. Whereas in western countries protease inhibitors is used as a part of regimen. Both protease inhibitors and nucleoside and non nucleoside reverse transcriptase inhibitors causes’ hyperlipidaemia. The Incidence rate of avascular necrosis have been reported to be 0.135% in the general population, although incidence rates ranging 0.3-0.45% have been observed in human immuno deficiency virusinfected patients. [3].The femoral head is the main location of avascular necrosis and is bilateral in approximately 80% of the cases. Other sites of avascular necrosis involved are knee, ankle, shoulder and wrist. [4, 5] Previously, avascular necrosis has been infrequently described in human immuno deficiency virus -infected patients; but in recent reports, its incidence in these patients has grown between 45–58 times over the rate expected in the general population. [6, 7] A retrospective chart review conducted from 19 human immuno deficiency virus clinics in Spain reported 23 cases of avascular necrosis over a 10- year period with a notable increase in frequency of avascular necrosis from 1.6 per 1,000 AIDS patients during 1993-1996 to 14 per 1,000 patients during 1997-2000. [1] Elevation of cholesterol has been observed with the use of efavirenz and nucleoside reverse transcriptase inhibitors such as stavudine. [8, 9] More recently, higher triglycerides levels were reported in subjects taking stavudine, compared with tenofovir-based regimen. [10] A prospective, randomized, double blind study assessed 602 antiretroviral-naive patients who received tenofovir or stavudine, in combination with lamivudine and efavirenz, and who were observed for 144 weeks. [10] A significant increase in the triglyceride level was observed among stavudine-treated patients. [10] Osteonecrosis was seen in patients with different levels of immune status. In most patients, CD4 counts at diagnosis of osteonecrosis were above 200 cells/mm3 . In some of the reported cases, although the average CD4 count was 501 cells/mm3 and the human immune viral RNA was suppressed (<50 cell/mm3 ), many patients still developed avascular necrosis.[1] In our case, CD4 level was 590 cells/mm3 . The mechanisms by which highly active antiretro viral therapy could cause osteonecrosis are unclear, but they involve increased production of anti–protein S or antiphospholipid antibodies as a result of enhanced humoral immunity and hyperlipidaemia. [4] Alteration in lipid metabolism is one of the mechanisms even involved in glucocorticoid-associated osteonecrosis. [10] Osteonecrosis is reported to occur most often in association with advanced human immuno deficiency virus disease. [2] Moreover, in most of the human immuno deficiency virus infected patients, known predisposing factors are lacking, which suggests a possible role of antiretroviral therapy. [3] Till date avascular necrosis of the bone due to anti retroviral therapy is not yet reported from India.
CONCLUSION
In western countries, protease inhibitors are commonly used in multiple drug regimens. All the reported cases of avascular necrosis due to anti retro viral therapy are mainly focusing the role of protease inhibitors causing avascular necrosis of femoral head. Whereas in India, nucleoside and non nucleoside reverse transcriptase inhibitors are used as per National Aids Control Organisation (NACO) as the main drug in combination therapy without protease inhibitors. We want to enlighten the medical fraternity that first line of drugs like stavudine, lamivudine and efavirenz used as per Indian National AIDS Control Organisation (NACO) is known to cause avascular necrosis as in our case. We report this case report for the following reasons. 1. The first case of avascular necrosis of the bone in India due to anti retroviral therapy. 2. To highlight the significance of screening the hips in the MRI study of lumbo sacral spine in ART therapy patients with low back pain.

References:
REFERENCES
1. Ramani Reddy MD, Monika ND, Robert D, Arjun Dutta, Jacquay Oliver, and Winston Frederick. Avascular Necrosis and Protease Inhibitors. Journal of the National Medical Association. November 2005; 97: no.11.1543- 1546.
2. Paul Monier, Kevin McKown, Michael S. Bronze. Osteonecrosis Complicating Highly Active Antiretroviral Therapy in Patients Infected with Human Immunodeficiency Virus.CID 2000; 31 (December).1488-1492
3. Fe´lix Gutie´rrez, eT al. Osteonecrosis in Patients Infected with HIV: Clinical Epidemiology and Natural History in a Large Case Series from Spain .J Acquir Immune Defic Syndr .July 2006; Vol 4:No3.
4. Aurélie C Molia, Christophe Strady, Christine Rouger, Isabelle M Beguinot, Jean-Luc Berger, and Thierry C Trenque. Osteonecrosis in Six HIV-Infected Patients Receiving Highly Active Antiretroviral Therapy. The Annals of Pharmacotherapy. 2004 December; Volume 38:2050-2054.
5. Gerster JC, Camus JP, Chave JP, et al. Multiple site avascular necrosis In HIV infected patients. J Rheumatol 1991; 18:300– 2.
6. Wolfe Cj, Taylor-Butler Kl. avascular necrosis. A case history and literature review. Arch FAM Med. 2000; 9:291–294.
7. Mankin HJ. Nontraumatic necrosis of bone (osteonecrosis) N Engl J Med. 1992; 326:1473–1479.
8. Calza L, Manfredi R, Mastroianni A, Osteonecrosis and highly active antiretroviral therapy during HIV infection: report of a series and literature review. AIDS Patient Care STDS. 2001 Jul; 15(7):385-9.
9. Hamilton D, Rivaldo VC and Anamaria MM Paniago. Dyslipidaemia Associated with the Highly Active Antiretroviral Therapy in AIDS Patient: Reversion after Switching (Stavudine to Tenofovir and Lopinavir/Ritonavir to Atazanavir/Ritonavir). The Brazilian Journal of Infectious Diseases 2007; 11(2):290-292.
10. Gallant JE, Staszewski S, Pozniak AL, et al. Efficacy and safety of tenofovir DF vs. Stavudine in combination therapy in antiretroviral-naïve Patients: a 3-year randomized trial. JAMA 2004; 292:266–8.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License