IJCRR - 7(14), July, 2015
Pages: 01-06
Print Article
Download XML Download PDF
SYNTHESIS AND EVALUATION OF ANTIOXIDANT ACTIVITY (IN-VITRO) OF SOME N-PHTHALYL-[{(3-NITRO ANILINO)-4\" ARYL SUBSTITUTED}]-1, 3, 4 THIADIAZOLES
Author: Pramilla Sah, Kuldeep Sen
Category: Healthcare
Abstract:1, 3, 4- thiadiazoles are potential bioactive agents having the toxophoric N=C\?S linkage. There are several isomers of thiadiazole, i.e.1,2,3-thiadiazole, 1,2,4-thiadiazole, 1,2,5-thiadiazole, and 1,3,4-thiadiazole. However 1, 3, 4-Thiadiazole is the main isomer of the thiadiazole series which has versatile pharmacological activities. Hence in this study we have synthesized some new substituted 1, 3, 4 thiadiazoles i.e. N-phthalyl-[{(3-nitro anilino)-4'' aryl Substituted}]-1, 3, 4 thiadiazoles. The title compounds were prepared from N-phthayl-3-nitro anilino thiosemicarbazide by treating N-phthayl-3-nitro aniline with carbon disulphide,
ammonium hydroxide and hydrazine hydrate. The synthesized compounds were then evaluated for their in- vitro antioxidant activity by determining the reducing power and hydrogen peroxide scavenging potential. The results show that one derivative V2 (N-Phthalyl-[{(3'-nitro anilino)-5-(4''-chloro phenyl)}]-1, 3, 4-thiadiazole) had the best redusing ability while V4 (N-Phthalyl-[{(3'- nitro anilino)-5- (4''-bromo phenyl)}]-1, 3, 4-thiadiazole) was the strongest oxidant. The chemical structures of the synthesized compounds have been characterized by IR, 1HNMR and mass spectra.
Keywords: N-phthalyl, 1, 3, 4-thiadiazoles, Antioxidant activity
Full Text:
INTRODUCTION
There is a vast variety of heterocyclic compounds which have medicinal importance. Recently, much attention has been focused on thiadiazole derivatives in view of their broad spectrum of activities as thiadiazoles have much importance in medicine, agriculture and technology. A large number of 1, 3, 4-thiadiazoles have been patented in the agricultural field as herbicides and bactericides. The N−C=S linkage is an essential structural feature which is responsible for organic sulphur containing pesticides such as dithiocarbamates, thioureas and thiosemicarbazides. Literature survey has shown this nucleus to exhibit diverse activities i.e. antimicrobial1-4, antifungal5 , anti-inflammatory 6-8, antileishmanial activity9 , anticancer activity, 10-11 anti-HIV activity12, antioxidant/radioprotective activity-13-14, anti-Helicobacter pylori activity15etc. Hence some new 1, 3, 4,-thiadiazole derivatives have been synthesized having this lead pharmacophore to investigate their antioxidant activity.
MATERIALS AND METHODS
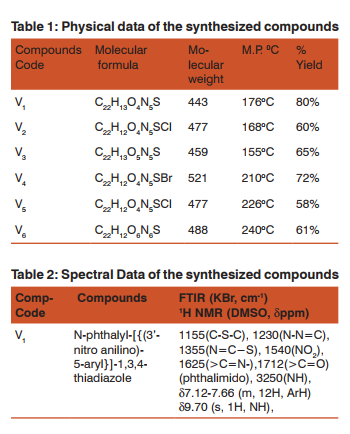
Melting point of the synthesized compounds was taken in a ‘Neolab’ electrical apparatus. All the synthesized compounds were characterized by analytical and spectral techniques of IR, NMR and Mass spectroscopy (Table1-2). FTIR was carried out on Varian and Brucker models, 1H’NMR on Brucker 400 with DMSO as the solvent while Mass was recorded on JEOL-SX-102/DA6000 mass spectrophotometer instrument using Xenon (6KV,10MA) as the FAB gas. U.V. visible (U.V.-2601) of Rayleigh was used to evaluate the antioxidant activity (in- vitro).
Procedure
Synthesis of N - phthalyl-3-nitro aniline (III) Equimolar amount of phthalimide and 4-chloro-2-nitro aniline, in presence of (0.002 mol) of anhydrous potassium carbonate were refluxed in dry acetone (10mL) for 5-6 hrs. on a water bath. The residual potassium carbonate was filtered off. Filterate was concentrated, cooled and solid thus obtained was filtered, dried and recrystallized from ethanol. Synthesis of N-phthalyl-(3-nitro anilino)-thiosemicarbazide (IV) 16 0.02 mole of N-phthayl-3-nitro aniline was taken in a round bottom flask and 30mL absolute ethanol was added to it. In a separate beaker 0.02 mole of carbon disulphide was added to 20 mL ammonium hydroxide. This mixture was added slowly drop wise to the ethanolic solution with constant magnetic stirring for over half an hour. A clear solution was obtained. This was kept at room temperature for 1hr. Then 15mL hydrazine hydrate (10%) was added to the above reaction mixture again with constant stirring. A brown colored solution obtained was concentrated to half of its original volume and left overnight in a refrigerator. The solution was acidified using concentrated HCl and white precipitate obtained was washed with cold water filtered, dried and recrystallized with absolute ethanol.
Synthesis of N-phthalyl-[{(3’-nitro anilino)-5-aryl}]-1, 3, 4 thiadiazole (V1)
Equimolar ratio of N-phthayl-(3-nitro anilino) thiasemicarbazide intermediate synthesized in the previous step and benzoic acid was taken in a round bottom flask. The contents were then heated on a sand bath for approx. 4hr. without any solvent under moisture free condition. The fused material was then treated with 10% cold solution of NaHCO3 . A solid mass separated out was dried, recystallized from absolute ethanol after filtration. Other compounds (V2 -V6 ) were synthesized following the same procedure with different aryl substituted aromatic carboxylic acids namely p-chloro, p-hydroxy, p-bromo, ochloro and o-nitro respectively.
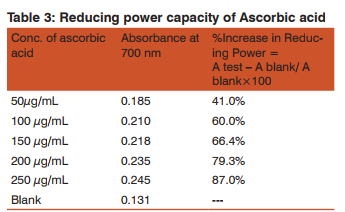
Antioxidant activity: Reducing power by FeCl3 17 Preparation of standard solution: 10mg of ascorbic acid was dissolved in 10 ml of water. Dilution of this solution was done with distilled water to give concentrations of 50 µg/mL, 100 µg/mL, 150 µg/mL, 200 µg/mL and 250 µg/mL respectively.
Phosphate buffer: 0.2M phosphate buffer of pH 6.6 was prepared according to IP. Preparation of test samples: Required quantities of the test sample were dissolved in minimum quantity of methanol and volume was made up to 10ml with phosphate buffer.
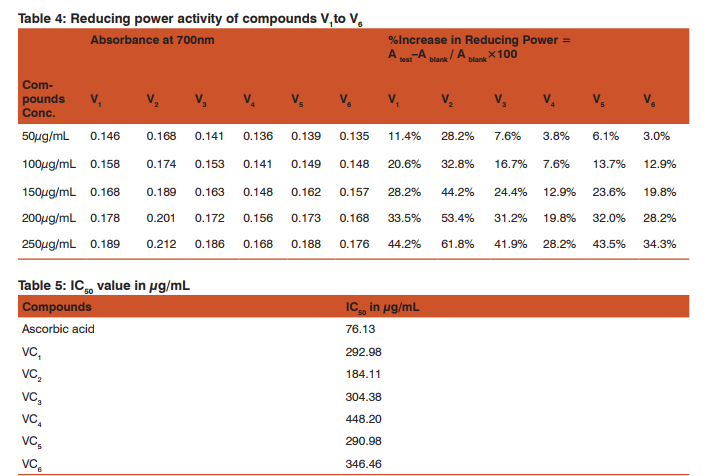
Procedure: 2.0mL of each sample and standard solution were mixed with 2.5mL of 1% potassium ferricyanide solution. The mixture was incubated at 50o C for 20 min. and after cooling aliquots of trichloroacetic acid 2.5mL were added to the mixture and the solution was left for 10 min. Then 2.5 ml of each sample was mixed with 2.5mL of distilled water, 2.5mL of freshly prepared ferric chloride solution. After 10min, absorbance was measured at 700 nm. A blank was prepared without adding the sample. Ascorbic acid at various concentrations was used as the standard. A test −A blank % Increase in Reducing Power = × 100 A blank Where; A test = Absorbance of test solution A blank = Absorbance of blank Standard = Ascorbic acid. The antioxidant activity of the sample compounds was compared with the standard i.e. ascorbic acid. Each experiment was carried out in triplicate.
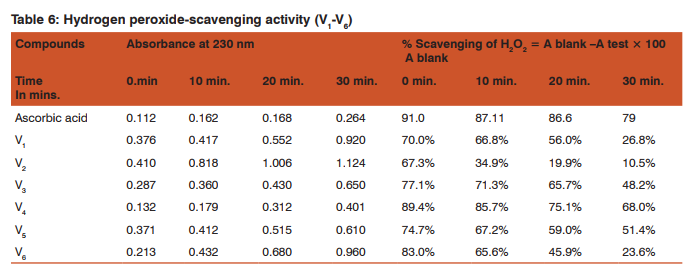
Hydrogen Peroxide-scavenging activity18 0.1M Phosphate buffer (pH 7.4): 0.1M phosphate buffer of pH 7.4 was prepared according to IP. 40 mM solution of hydrogen peroxide: 4.535 mL of given 30%(w/v)H2 O2 was dissolved in 1000mL of distilled water to give 0.04M or 40mM solution of H2 O2 .
Preparation of test samples: Required sample 10mg was dissolved in minimum quantity of methanol and volume was made up to 10ml with phosphate buffer. Separately all the samples were diluted in 10mL volumetric flask with phosphate buffer and 1mL was made up to 10mL to give a concentration of 100µg /mL
Procedure: The hydrogen peroxide-scavenging activity of synthesis compound was determined by the method of Ruch et al.18 The compounds were dissolved in 3.4 mL of 0.1M phosphate buffer (pH 7.4) and mixed with 600 µL of 40 mM solution of hydrogen peroxide. The absorbance value at 230 nm of the reaction mixture was recorded at 10 min intervals between zero and 30 min. for each concentration. Separate blank sample was used for back ground substraction. The percentage of hydrogen peroxide scavenging activity was determined as follows: A blank − A test %Scavenging H2 O2 = ×100 A blank
RESULTS
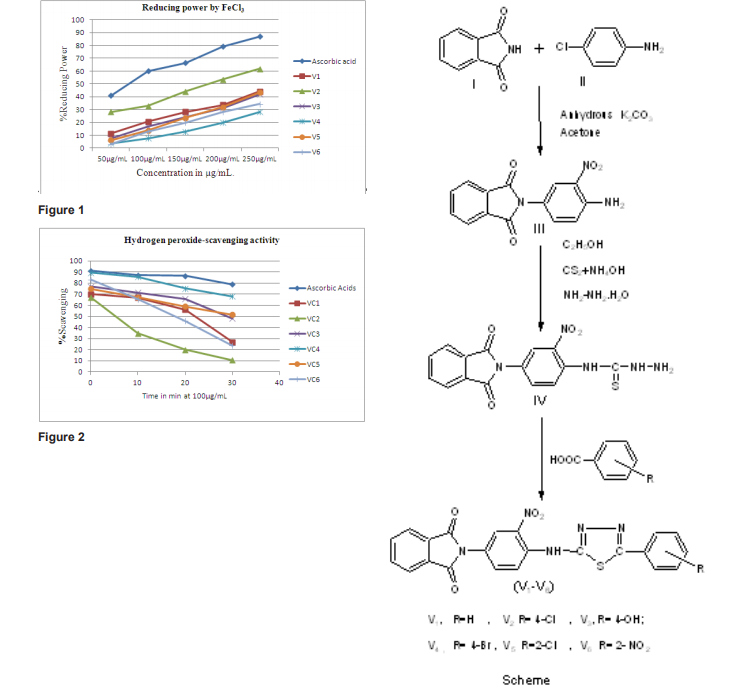
Table 3 and 4 depict the reducing power of derivatives V1 -V6 by FeCl3 with ascorbic acid as the standard. Table 6 shows the oxidizing potential using H2 O2 while table 5 gives the IC50 values of the derivatives with respect to ascorbic acid.
DISCUSSION
Reducing power by FeCl3
At 250µg/mL, V1 has 44.2% increases in the reducing activity while V5 has 43.5%. The IC50 data also shows this similarity as values approx. 293 is obtained for V1 and 291 for V5 . However derivative V2 has 61.8% (Table 4) reducing ability at a concentration of 250µg/mL and is the most active among this series. Ascorbic acid also shows 60% reducing power though at a lower concentration of 100µg/mL(Table 3). Hence large dose of V2 will be required to produce the same effect. An IC50 value is also least in this compound. (Table 5) Derivatives V 1 and V5 also show similar behavior with 33.5% at 200µg/mL(V1 ) and 32%(V5 ) at the same concentration. Hence the above observations show V2 has the best reducing ability while derivative V4 is the least reductant. Structure activity relationship shows all these derivatives have an NH group linked to the phthalyl and thiadiazole ring which might be one factor for the reducing capability of these compounds.
Oxidizing Power
Maximum hydrogen peroxide scavenging activity is observed in V4 (89.4%) at 0 min. and 85.7% at 10min. comparable to ascorbic acid 91% (0min) and 87% after 10min. This is in agreement to the reducing power which is the least. Among V5 and V6 initially V5 has better activity 83% at 0 min. but with time the value falls to 23.6%. However in derivative V5 the oxidizing power shows a gradual decline from 74.7% to 51.4% in a span of 0 to 30 min. thus being better oxidant. Similar pattern is visible in derivative V3 with values between77.1% to 48.2% are obtained in the same time period. It can hence be concluded that V4 is the must active compound while V2 is the least. The behavior of V2 as the weakest oxidant matches with its reducing power ability which is the highest. (Table 6) Presence of substituents like nitro and hydroxyl group attached to the phthalyl substituted thiadiazole ring might be responsible for the oxidizing potential of these compounds.
CONCLUSION
Out of the six derivatives tested for in-vitro antioxidant activity maximum reducing power has been observed in compound V2 , while V4 has shown the highest oxidizing ponential. This is in agreement to the result observed as V2 has the least oxidizing power while V4 is the least reductant.
ACKNOWLEDGMENT
The authors are thankful to Head Department of Chemistry J.N.V. University, Jodhpur, for provided all the laboratory facilities. Thanks are also to the Director of (S.A.I.F.), Punjab University Chandigarh, for the spectral analysis.

References:
1. Mohan J, Rathee A. synthesis and antimicrobial activity of s-triazolo[3,4-b][1,3,4]thiadiazoles, s-triazolo[3,4-b] [1,3,4] thiadiazines and s-triazolo. Ind J Hetero Chem 2011; 21: 117-120.
2. Demirbas A, Sahin D, Demirbas N, and Karaoglu SA. Synthesis of some new 1,3,4-thiadiazol-2-ylmethyl-1,2,4-triazole derivatives and investigation of their antimicrobial activities. Euro J Med Chem 2009; 44(7): 2896–2903.
3. Hussain S, Sharma J, Amir, M. Synthesis and antimicrobial activities of 1,2,4-triazole and 1,3,4-thiadiazole derivatives of 5-amino-2-hydroxybenzoic acid. E-J. Chem 2008; 5(4):963–968.
4. Lamani RS, Shetty NS, Kambl RR, Khazi AM. Synthesis and antimicrobial studies of novel methylene bridged benzisoxazolyl imidazo[2,1-b][1,3,4]thiadiazole derivatives. Euro J Med Chem 2008; 44:2828–2833.
5. Chen CJ, Song BA, Yang S. et al., Synthesis and antifungal activities of 5-(3,4,5-trimethoxyphenyl)-2-sulfonyl-1,3,4- thiadiazole and 5-(3,4,5-trimethoxyphenyl)-2-sulfonyl1,3,4-oxadiazole derivatives. Bioorg Med Chem 2007;15: 3981–3989.
6. Jyothi C, Das AK, Pal P, Baidya M. synthesis and anti-inflammatory activities of some new substituted thiadiazolothieno- pyrimidinones. Indian J Hetero Chem 2007; 17: 191-192.
7. Karegoudar P, Prasad DJ, Ashok M, Mahalinga Poojary MB, Holla BS. Synthesis, antimicrobial and anti-inflammatory activities of some 1,2,4-triazolo[3,4-b][1,3,4] thiadiazoles and 1,2,4-triazolo[3,4-b][1,3,4]thiadiazines bearing trichlorophenyl moiety. Euro J Med Chem 2008; 43(4):808–815.
8. Goksen US, Kelekci NG, Goktas O. et. al., 1-Acylthiosemicarbazides, 1,2,4-triazole-5(4H)-thiones, 1,3,4-thiadiazoles and hydrazones containing 5-methyl-2-benzoxazolinones: synthesis, analgesic-anti-inflammatory and antimicrobial activities. Bioorg Med Chem 2007; 15(17): 5738–5751.
9. Behrouzi-Fardmoghadam M, Poorrajab F, Ardestani SK, Emami S, Shafiee A, Foroumadi A. Synthesis and in vitro anti-leishmanial activity of 1-[5-(5-nitrofuran-2-yl)-1,3,4- thiadiazol-2-yl]- and 1-[5-(5-nitrothiophen-2-yl)-1,3,4-thiadiazol-2-yl]-4-aroylpiperazines. Bioorg Med Chem 2008; 16(8): 4509–4515.
10. Wei MX, Feng L, Li XQ, Zhou XZ, Shao ZH, Shao H. Synthesis of new chiral 2,5-disubstituted 1,3,4-thiadiazoles possessing γ-butenolide moiety and preliminary evaluation of in vitro anticancer activity. Euro J Med Chem 2009;44 (8): 3340–3344.
11. Ibrahim DA. Synthesis and biological evaluation of 3,6-disubstituted [1,2,4]triazolo[3,4-b][1,3,4]thiadiazole derivatives as a novel class of potential anti-tumor agents. Euro J Med Chem 2009;44:2776–2781.
12. Zhan P, Liu X, Fang Z, Li Z, Pannecouque C, Clercq ED. Synthesis and anti-HIV activity evaluation of 2-(4-(naphthalen-2-yl)-1,2,3-thiadiazol-5-ylthio)-N-acetamides as novel non-nucleoside HIV-1 reverse transcriptase inhibitors. Euro J Med Chem 2009; 44: 4648–4653.
13. Kus C, Kilcigil GA, Ozbey S. et al., Synthesis and antioxidant properties of novel N-methyl-1,3,4-thiadiazol-2-amine and 4-methyl-2H-1,2,4-triazole-3(4H)-thione derivatives of benzimidazole class. Bioor Med Chem. 2008;16: 4294– 4303.
14. Cressier D, Prouillac C, Hernandez P. et al., Synthesis, antioxidant properties and radioprotective effects of new benzothiazoles and thiadiazoles. Bioorg Med Chem. 2009; 17(14): 5275–5284.
15. Foroumadi A, Rineh A, Emami S. et al., Synthesis and antiHelicobacter pylori activity of 5-(nitroaryl)-1,3,4-thiadiazoles with certain sulfur containing alkyl side chain, Bioorg Med Chem Lett. 2008; 18(11):3315–3320.
16. Sah P, Parmar S, Synthesis and Insecticidal Activity of Some New 2’-[1-Amino Methyl-{N-Alkyl phthalyl}- Benzimidazolo]-5’-(2’’/4’’/3’’/5’’-Aryl Substituted)-1’, 3’,4’-Thiadiazoles. Ultra Chem 2010:6(2):199-210.
17. Oyaizu M. Studies on product of browing reaction prepared from glucose amine. Jpn. J. Nutr 1986; 07: 307-315.
18. Ruch RT, Cheng SJ, Klaunig JE. Spin trapping of superoxide and hydroxyl radicals methods in enzymology 1984; 105: 198-209.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License