IJCRR - 8(19), October, 2016
Pages: 17-20
Print Article
Download XML Download PDF
SEROPOSITIVITY OF HEPATITIS B VIRUS AND HEPATITIS C VIRUS AMONG BLOOD DONORS AT A TERTIARY CARE HOSPITAL IN KASHMIR: A TEN-YEAR STUDY
Author: Mohammad Zubair Qureshi, Humaira Bashir, Peer Maroof, Samoon Jeelani, Nazia Bhat, Fehmeeda Akhter
Category: Healthcare
Abstract:Objectives: Hepatitis B virus (HBV) and Hepatitis C virus (HCV) are important transfusion-transmissible infections. This study was performed to assess the Seropositivity of HBV and HCV Seropositivity among blood donors at a Tertiary Care Hospital based blood bank at SKIMS, Kashmir.
Materials and Methods: The blood donation records over 10 years from Jan- 2003 to Dec-2012 were reviewed, retrospectively, for the seropositivity and yearly trends of HBV and HCV.
Results: A total of 97427 donations were received. The overall number of HBV seropositive donations was 469 and that for HCV was 194, with the seroprevalence rates of 0.48% for hepatitis B surface antigen (HBsAg) and 0.20% for HCV. The seropositivity rate was higher in the replacement donors compared to the voluntary donors and 0% in repeat voluntary donors. The annual rates showed decreasing trends for both HBsAg and HCV.
Conclusion: The seropositivity of viral markers was lower in our study as compared with studies from other parts of the country. It was significantly lower in voluntary donors compared to replacement donors and repeat voluntary donors were found to be much safer.
Keywords: Hepatitis B virus, Hepatitis C virus, Seropositivity, Hepatitis B Surface Antigen
Full Text:
INTRODUCTION
Hepatitis B virus (HBV) and Hepatitis C virus (HCV) are worldwide healthcare problems, especially in developing countries. It is estimated that approximately one third and 3% of the global population has been infected with HBV and HCV, respectively.1, 2 Viral hepatitis, caused by HBV and HCV is one of the common transmissible cause of chronic liver morbidity and mortality. The main modes of transmission include blood transfusion, unsafe sex, use of parental drugs, and the vertical transmission from mother to child. Blood transfusion contribute to the ever widening pool of these infection, wherein even an asymptomatic person (carrier) can transmit the infection.3
Initially, progression of blood-borne infections led the scientists to design method of prevention. Characterization of transfusion-transmitted pathogens, development of strategies to measure infection rates in blood donors as well as in recipient populations, characterization of the early viremia, and implementation of more restrictive donor eligibility criteria and increasingly sensitive laboratory techniques for donor screening have made blood supplies be safe in developed countries.4,5 In developing countries, the prevention and determination of blood borne infections have not been well improved yet. So, transfusion as a main way for blood-borne infections continues to cause serious problems in developing countries and less serious in developed countries.6
Screening and assessment of blood donors not only alleviates the risk of transmission through infected blood products, but also gives an idea about the prevalence rates of the infections in the community. This cross sectional observational study was carried out in the department of Transfusion Medicine, SKIMS (Srinagar, Jammu and Kashmir, India) a Tertiary Care Hospital. This study was aimed to assess the seropositivity of the markers of HBV and HCV in the blood donors at the blood bank over the period of 10 years.
MATERIALS AND METHODS
The blood donation records over 10 years (2003-2012) were reviewed retrospectively, for a total of 97427 donors, ranging from 18-60 years age. The hospital has a policy of using a standard blood donor screening questionnaire and excluding the high-risk donors. Replacement donations (from family, relatives and friends of the patient) accounted for the majority of the donations while the voluntary donors formed the rest.
Third generation ELISA kits were used for detection of HBV and HCV. HBsAg ELISA Test kit, manufactured by In Tec Products, INC. The advanced HBsAg test kit is an enzyme-linked immunosorbent assay (ELISA) for the qualitative determination of Hepatitis B Surface Antigen (HBsAg) in human serum or plasma. HCV detection was done Using Zhongshan HCV ELISA kit, manufactured by Zhongshan Bio-tech Co, Ltd. This test is enzyme-linked immunosorbent assay (ELISA) for the qualitative determination of antibody to Hepatitis C virus in human plasma. All the donors who turned out to be reactive for HBsAg and HCV initially were subjected to reconfirmation by testing them again twice on consecutive days. The blood that was still seropositive was discarded. All the donors who turned out to be positive for viral markers were notified and counselled about this via telephone/mobile calls or letters and were requested to come to our hospital for further medical evaluation and possible treatment.
Data was collected, coded, entered and analysed using Statistical Package for Social Sciences (SPSS), Version 17 and Minitab Packages. Data was expressed as percentage and the respective comparisons were made by Chi-square test. Seropositivity rates were calculated and a p value of less than 0.05 was taken to be significant.
RESULTS
Total of 97427 donations were received over a period of 10 years (Jan 2003- Dec 2012). Of total donations 95258 (97.8%) were males and 2169 (2.2%) were females. Maximum donors 46181 (47.4%) were in the age group of 18-30 years, 25234 (25.9%) were between 31-40 years, 16757 (17.2%) were between 41-50 years and 9255 (9.5%) were between 51-60 years. The replacement donations comprised 94406 (96.9%) while voluntary donations were 3021 (3.1%) of total donations. First time voluntary donors, which had no history of previous voluntary blood donation were 2018 (66.8%) and repeat voluntary donors which had history of one or more previous voluntary blood donations were 1003 (33.2%).
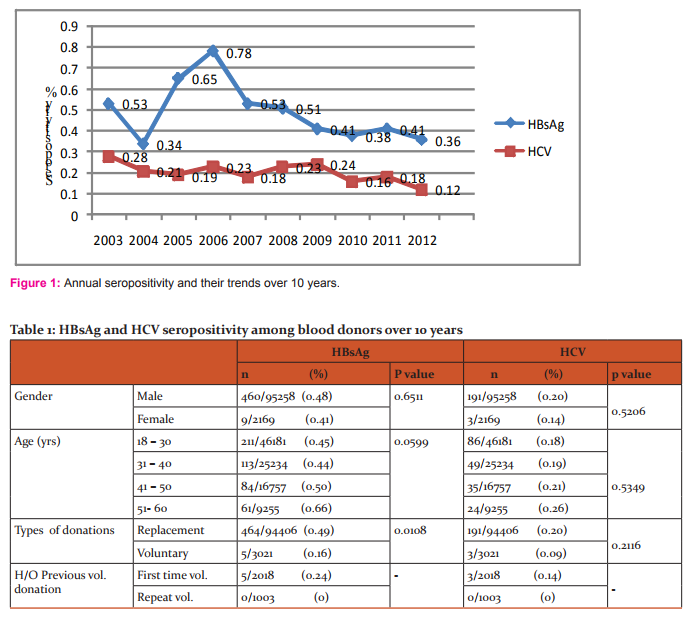
The number of HBsAg seropositive donations was 469 and that for HCV was 194, with the Seropositivity rates of 0.48% for HBsAg and 0.20% for HCV. The annual Seropositivity and their trends over 10 years are shown in Figure 1.
The Seropositivity of HBsAg and HCV on the basis of Gender, Age, and Type of donation and H/O previous voluntary donation is depicted in Table1.
DISCUSSION:
Every blood transfusion carries a potential risk for transmissible disease.7 This reflects the need and importance of the mandatory screening of the above infectious markers in blood donations. The prevalence of infection among blood donors has been used as a surrogate marker for the prevalence of infection in the population at large. Although certain pitfalls, like the exclusion of people below 18 years and over 60 years and low number of female donors have been cited, it is still an important indicator of the disease burden.
HBV and HCV are important transfusion transmissible diseases. The HBsAg testing on the donated blood has been done for several years now, but in our country, compulsory testing for HCV came into full effect only after 2002.8 HBV is one of the major global public health problems. HBV infection is the 10th leading cause of death and HBV-related hepatocellular carcinoma is the fifth most frequent cancer worldwide. Approximately 30% of the world’s population has serologic evidence of current or past infection with HBV. India lies in an intermediate HBV endemicity zone and the number of HBV carriers is estimated to be 50 million, forming the second largest global pool of chronic HBV infections.9
HBV prevalence in general population in India is 2% to 8% and 1% to 2% in the blood donors, according to various studies.9-11 Panda and Kar11 did a study in Orissa, a state in the eastern part of the country, and reported the prevalence of HBsAg in blood donors to be 1.13%. Pahuja and colleagues12 reported the prevalence to be 2.23% in Delhi. Garg and co-workers13 reported that the prevalence was as high as 3.44% in the western part of the country.
In our study, the overall seropositivity for HBV over 10 years was 0.48%. The seropositivity ranged from 0.53% in 2003 to 0.36% in 2012, showing decreasing trends. The overall seropositivity rate in males was 0.48% while in females it was 0.41%. The seropositivity rates of hepatitis B were lower among young donors between 18-30 years of age (0.45%), as compared to older donors between 51-60 years of age (0.66%). This may be explained on the basis of increased exposure with age and on the fact that a high awareness of blood-borne viral infections has developed and a comprehensive vaccination program against hepatitis B has been implemented. The seropositivity in voluntary donors was significantly lower compared to replacement donors (0.16% vs. 0.49%) and 0% in repeat voluntary donors emphasizing the fact that donor recruitment is vital and repeat voluntary donations should be encouraged to ensure more safe blood supply.
HCV infection is an evolving public health problem globally. This virus infects approximately 3% of the world population; placing approximately 170 million people at risk for developing HCV related chronic liver disease.14 The global seroprevalence of HCV among blood donors varies from 0.4% to 19.2%. However, overall HCV prevalence in India has been reported to be less than 2%. The seroprevalence of HCV in voluntary blood donors in India is between 0.12 and 2.5%. 10-13 In two studies performed in different parts of Delhi, the prevalence of HCV in blood donors has been reported to range from 0.66% to 2.5%.12,15 Garg and co-workers13 reported the HCV prevalence in blood donors in western India to be 0.28%. In Orissa, the HCV prevalence was reported to be 1.98% by Panda and coworkers.11
In our study, the overall 10 year HCV seropositivity was 0.20%. The seropositivity of HCV infection among blood donors in the past 10 years showed a decreasing trend, from 0.28 in 2003 to 0.12 in 2012. Seropositivity of HCV was seen more in males (0.20%) as compared to females (0.14%). Seropositivity of HCV was found higher in the age group of 51-60 years (0.26%), with the lowest seropositivity in age group of 18-30 years (0.18%). Beenu et al16 in their study found that the HCV seropositivity increased with age in voluntary donors, with the lowest prevalence in the age group of 18-30 years, and highest in the age group of 51-60 years. This was comparable with one study. The prevalence seems to increase with age either because of a continuing risk of exposure or a cohort effect with declining risk in more recent time. In our study, seropositivity of HCV in voluntary donors was significantly lower compared to the replacement donors (0.09% vs. 0.20%) and 0% in repeat voluntary donors again emphasizing that repeat voluntary donations should be encouraged.
CONCLUSION
The seropositivity of viral markers was lower in our study as compared with studies from other parts of the country. It was lower among younger age groups and significantly lower in voluntary donors compared to replacement donors and repeat voluntary donors were found to be much safer. Stringent measures need to be taken including, dissemination of information, better donor recruitment and retention strategies, promoting repeat voluntary blood donations and strict screening of blood and blood products to increase the blood safety.
ACKNOWLEDGEMENTS:
Authors acknowledge the immense help received from the scholars whose articles are cited and included in references of this manuscript. The authors are also grateful to authors / editors / publishers of all those articles, journals and books from where the literature for this article has been reviewed and discussed.
FINANCIAL SUPPORT: None
CONFLICT OF INTEREST: None
References:
1. Rezvan H, Abolghassemi H, Kafiabad SA. Transfusion transmitted infections among multitransfused patients in Iran: a review. Transfuse Med 2007; 17(6):425–33.
2. Livramento A, Cordova CM, Spada C, Treitinger A. Seroprevalence of hepatitis
B and C infection markers among children and adolescentsin the southern Brazilian region. Rev Inst Med Trop Sao Paulo 2011; 53(1) :13-7.
3. Simmonds P.The origin and evolution of hepatitis viruses in humans. J Gen Virol 2001;82:693-712.
4. YeeTT, Lee CA. Transfusion transmitted infection in hemophilia in developing countries. Semin Thromb Hemost. 2005; 31(5):527-37.
5. Samimi-Rad K , Shahbaz B. Hepatitis C virus genotypes among patients with thalassemia and inherited bleeding disorders in Markazi province, Iran. Haemophilia 2007;13(2):156-63.
6. Burki MF, Hassan M, Hussain H, Nisar Y, Krishan J. Prevalence of anti-hepatitis C antibodies in multiply transfused beta thalassemia major patients. Ann Pak Inst Med Sci 2005;1:150-3.
7. Kleinman SH, Lelie N, Busch MP. Infectivity of human immunodeficiency virus-1, hepatitis C virus, and hepatitis B virus and risk of transmission by transfusion. Transfusion 2009;49:2454-89.
8. Mukhopadhyaya A. Hepatitis C in India. J Biosci 2008;33:465-73.
9. Datta S. An overview of molecular epidemiology of hepatitis B virus (HBV) in India. Virol J 2008;5:156.
10. Gupta N, Kumar V, Kaur A. Seroprevalence of HIV, HBV,HCV and syphilis in voluntary blood donors. Indian J Med Sci 2004;58:255-7.
11. Panda M, Kar K. HIV, hepatitis B and C infection status of the blood donors in a blood bank of a tertiary health care centre of Orissa. Indian J Public Health 2008;52:43-4.
12. Pahuja S, Sharma M, Baitha B, Jain M. Prevalence and trends of markers of hepatitis C virus, hepatitis B virus and human immunodeficiency virus in Delhi blood donors: a hospital based study. Jpn J Infect Dis 2007;60:389-91.
13. Garg S, Mathur DR, Garg DK. Comparison of seropositivity of HIV, HBV, HCV and syphilis in replacement and voluntary blood donors in western India. Indian J Pathol Microbiol 2001;44:409-12.
14. WHO. Global surveillance and control of hepatitis C. Report of a WHO consultation organized in collaboration with the viral hepatitis prevention Board, Antwerp, Belgium. J Viral Hepat 1999;6:35-47.
15. Sood G, Chauhan A, Sehgal S, Agnihotri S, Dilawari JB. Antibodies to hepatitis C virus in blood donors. Indian JGastroenterol 1992;11:44.
16. Beenu T, Nelam M, Chawla YK et al. Prevalence and significance of hepatitis C virus (HCV) seropositivity in blood donors. Indian J Med Res. 2006 (Oct.); 124:431-438.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License