IJCRR - 4(17), September, 2012
Pages: 59-66
Date of Publication: 14-Sep-2012
Print Article
Download XML Download PDF
AN IN VITRO SCREENING OF GROWTH INHIBITORY POTENTIAL OF ALLIUM SATIVUM TOWARDS SOME MICROBES OF SPOILAGE AND HEALTH SIGNIFICANCE
Author: Mamta Bhatia, Alka Sharma
Category: Healthcare
Abstract:The use of Allium sativum (garlic) as a cure and condiment predates written history. In present study aqueous extract, crude juice, essential oil and powdered form of Allium sativum were screened for their inhibitory potential towards some food borne pathogens, in culture media. Test microbes included : Bacillus cereus, Enterococcus faecalis, Escherichia coli, Psuedomonas aeruginosa, Psuedomonas alkaligenes, Shigella sonnei and Staphylococcus aureus. Spice agar method was opted for investigating
antibacterial activity of powdered spice samples. Agar well assay and broth dilution techniques were followed for determining growth inhibitory potentials of aqueous extract, crude juice and essential oil. Results revealed that essential oil most effectively inhibited bacterial strains followed by crude juice, while aqueous extract and powdered forms remained ineffective in arresting the growth of test bacteria.
Keywords: Antibacterial, Antimicrobial, Allium sativum, essential oil, garlic, spices
Full Text:
INTRODUCTION
Foods, by their very nature need to be nutritious and microbiologically stable . To ensure that food is safe and can be stored in a satisfactory state, it is necessary to either destroy the microorganisms present, or manipulate the food so that microbial growth is prevented or hindered. Resurgence in the use of natural herbal alternatives instead of synthetic preservatives to increase the shelf life of food commodoties has brought the use of aromatic plants to the forefront of investigations. Allium sativum, is one of the world?s most popular spices, and is used extensively from India to America, in French aioli, Greek skordalia, Indian korma, Turkish cacik and Vietnamese pho bo. Since ancient times, it has been used as a cure as well as food. Pliny the elder, a Roman naturalist, described in his Historia Naturalis how A. sativum could be used for gastrointestinal disorders, dog and snake bites, scorpion stings, asthama, madness, convulsions and tumours. Components from A. sativum modulate the cardiovascular and immune systems. Alongwith medicinal properties, it is known to have antiviral1,2 and antiprotozoal3,4 activities. Encouraged by these results, an in vitro trial was carried out to evaluate different forms of A. sativum viz. aqueous extract, crude juice, essential oil and powdered form, for their antimicrobial potencies, against seven food borne pathogens.
Materials and Methods
Procurement of spice samples
Fresh bulbs of A. sativum purchased in the amounts of 1 kg, from grocery shop, local market, Hisar, India. The spice samples were washed with clean water followed by distilled water to remove extraneous matter. The outer coverings of A. sativum bulbs/clove were peeled off manually with the help of knife. For the extraction of crude juice, peeled cloves of A. sativum were sliced into thin pieces and were crushed in pestle-mortar to get a thick paste. Thick pastes of spice samples were passed through sieve cloth. Filtrates thus obtained were sterilized by passing through syringe filter assembly having membrane filters of pore size 0.45 um under aseptic conditions. Crude extract thus obtained was stored in sterilized glass vials at 4+1° C and was used at various concentration levels within the 2 h. of their preparation. To get the powdered form , peeled cloves of A. sativum were dried in shade for 5 days followed by their grinding in the laboratory grinder and were kept in airtight containers till further use. Essential oil of A.sativum was procured from Aroma Chemicals Pvt. Limited, Delhi, India, stored in the dark amber colored, screw capped glass bottle and was kept away from light to avoid physicochemical changes in its composition. Purity of the essential oil was assured by the company to be more than 99.0 % .
Chemicals and culture media
Ethyl violet azide dextrose agar, Ethyl violet azide dextrose broth, MacConkey broth, MacConkey agar, Nutrient agar and Nutrient broth were obtained from HiMedia Pvt. Ltd, India. Dimethylsulphoxide (DMSO) and Sodium chloride (NaCl) were obtained from Central drug house Pvt. Limited, India.
Bacterial cultures
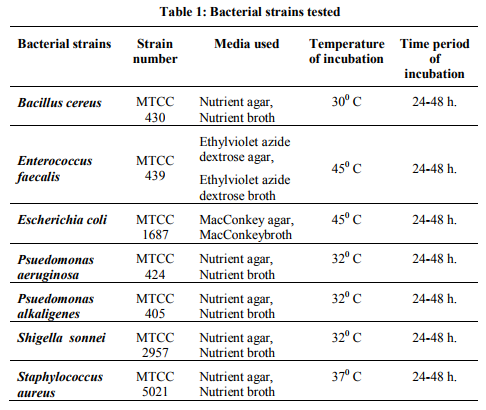
All the pure bacterial cultures viz. Bacillus cereus (MTCC 430), Enterococcus faecalis (MTCC 439), Escherichia coli (MTCC 1687), Psuedomonas aeruginosa (MTCC 1688), Psuedomonas alkaligenes (MTCC 405), Shigella sonnei (MTCC 2957) and Staphylococcus aureus (MTCC 5021) were obtained from Microbial Type Culture Collection (MTCC), Institute of Microbial Technology (IMTECH), Chandigarh, India. The reference bacterial strains were maintained on their respective media slants, subcultured bimonthly to maintain their viability and were stored at 4+1° C. Culture media, incubation temperatures and duration of incubation of reference bacterial strains are presented in Table 1.
Inoculum preparation
A flamed sterile wire loop was used to dislodge the lawns of test bacterial strains from their respective pure culture slants (24h. old) with 10 ml of sterilized normal saline (NaCl, 0.85% (w/v)) solution under aseptic conditions. Bacterial suspensions were adjusted with the same solution to contain approximately 1×107 cfu /ml and were utilized the same day.
Prepearation of aqueous extract
Aqueous extract of dried and powdered bulbs was prepared. Powdered spice sample was steeped overnight (temperature: 24-27°C) in sterilized distilled water in a ratio of 1:1 (w: v), followed by their homogenization in a blender at high speed for 2 min. The homogenized spice mixture was filtered through Whatmann No. 1 filter paper. Filtrate thus obtained, was sterilized by passing through syringe filters containing 0.45 um pore size membrane filters under aseptic conditions, collected in sterilized glass vial and was stored at 4+1° C. This aqueous extract was further used within the 2 h. of its preparation.
Preliminary screening of antibacterial activities of aqueous extract, crude juice and essential oil
Agar-well diffusion technique was followed5 . Freshly prepared inoculum (100 ul) of each reference bacterial strain was poured in plates with 20 ml of appropriate media. The petriplates seeded with bacterial strains were kept undisturbed for 30 min. for proper solidification and setting of agar to facilitate uniform digging of wells. Sterile cork borer (diameter: 8 mm) was used to bore wells in the solidified media plates previously seeded with bacterial inocula. Subsequently, different volumes of test substances were introduced into the wells of agar plates. Sterilized DMSO, instead of crude juice and essential oil served as negative control. These plates were allowed to stand at room temperature for at least 1 h. for the even diffusion of poured components and were incubated without inversion at their respective incubation temperatures in incubator for 24-48 h. After incubation, zones of inhibition formed around the wells were measured in millimeters (mm) and results were expressed as the net zone of inhibition which represented the subtraction of the diameter of the well (8 mm) from the measured zone.
Mnimum inhibitory concentrations (MIC) of crude juice and essential oil
MIC values of crude juice and essential oil were determined by broth dilution method6 . The media (broth) containing 2000 ul/ml of test substance was serially diluted twofold each with the media (broth) to give concentrations of 1000, 500, 250, 125, 62.50, 31.25, 15.62, 7.81, 3.90, 1.95, 0.97, 0.48, 0.24, 0.12, 0.06 ul/ml. Sterilized DMSO, instead of crude juice and essential oil, served as negative control. To the diluted solution, 100 ul of freshly prepared inoculum of each bacterial strain was added. These mixtures were incubated in the B.O.D. incubator at suitable incubation temperatures of microbes, for appropriate incubation periods. After the completion of incubation, 100 ul of the above mixture was evenly spread on the surface of solidified media petriplates with the help of sterile bent glass rod. These petriplates were incubated in an inverted position to observe the minimum concentration of test substances, at which visible growth of the reference microbes was completely inhibited.
Antibacterial activity of powdered form
Antibacterial activity of powdered form of spice sample was examined in culture media using spice agar method7 . Erelenmeyer flasks (100 ml capacity) containing 20 ml of appropriate media (containing agar) and powdered spice at different concentrations (0.1, 0.2, 0.4, 0.6, 0.8, 1.0, 1.5, 2.0, 2.5, 3.0, 3.5, 4.0, 4.5, 5.0, 5.5, 6.0 (%,w/v) were autoclaved at 121° C for 20 minutes. After autoclaving, spice agar mixtures (cooled but still molten) were poured into sterilized petriplates under aseptic conditions and these plates were kept undisturbed for 30 min. for proper setting of agar. Freshly prepared inoculum of each test microbe at 100 ul level was evenly spread over the entire surface of the respective solidified media in petriplate using a sterile bent glass rod. Seeded petriplates were incubated in incubator at appropriate temperatures and were examined for bacterial growth at 12 h. intervals, throughout the incubation period of 30 days. A similar experiment was carried out without any spice sample, that served as control. The time for initiation of microbial growth on control (without spice samples) and media supplemented with different concentrations of spice were recorded.
Statistical analysis :
All the experiments were performed in triplicates with two independent trials and the results obtained were highly reproducible. Values of growth inhibitory zones are mean ±SD (n=3) of three replicates.
RESULTS
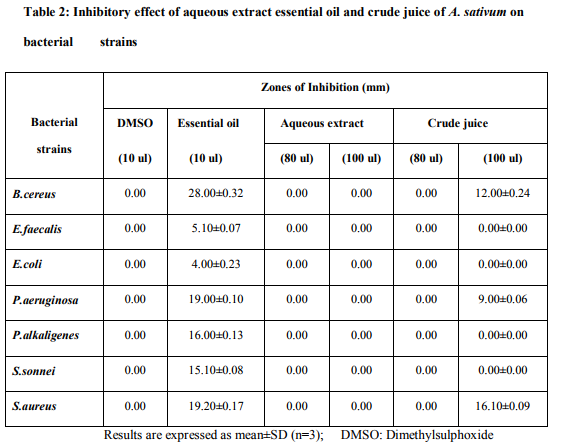
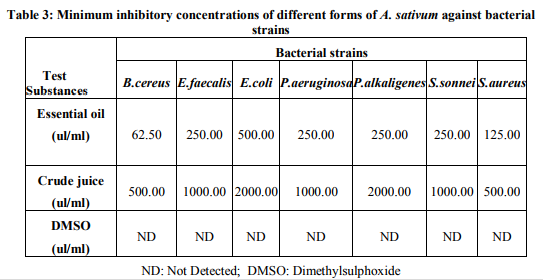
Zone inhibition assay results (Table 2) revealed that seeded petriplates with DMSO and aqueous extract of A. sativum , did not display growth inhibitory zones towards any bacterial strain under observation. Crude juice at 100 ul/well level exhibited inhibitory circles towards B. cereus, P.aeruginosa and S. aureus. On the other hand, essential oil of A. sativum, at 10ul/well exhibited distinct zones of inhibition towards all the bacterial strains under investigation. The diameter of inhibitory zones varied with the type of bacterial strains and test substances implicated in the study. Crude juice exhibited widest diameter of inhibitory zone towards S.aureus, while essential oil produced broadest inhibitory circle towards B.cereus. It was also observed that g+ve bacterial strains gave wider inhibitory zones towards test substances as compared to g-ve bacterial strains. On the basis of diameter of growth inhibitory zones, sensitivity of microbes in descending order towards test substances may be put in the following manner: Essential oil: B.cereus>S.aureus>P.aeruginosa>P.alkal igenes>S.sonnei>E.faecalis>E.coli. Crude juice: S.aureus>B.cereus>P.aeruginosa=E.coli =E.faecalis=P.alkaligenes=S.sonnei. Results of broth dilution technique depicted that crude juice and essential oil of A. sativum effctively inhibited all the bacterial strains (Table 3). MIC values of essential oil towards bacterial strains ranged from 62.50-500.00 (ul/ml), whereas, that of crude juice ranged from 125.00-500.00 (ul/ml), thereby indicating the higher susceptibility of the microbes towards former. It was noticed that higher concentrations of crude juice and essential oils were required to inhibit g-ve bacterial strains. Dried and powdered bulbs of A. sativum at all the concentration levels i.e. 0.0, 0.1, 0.2, 0.4, 0.6,0.8, 1.0, 1.5, 2.0, 2.5, 3.0, 3.5, 4.0, 4.5, 5.0, 5.5, 6.0 (%, w/v), remained ineffective in arresting bacterial strains, and visible growth of all the microbes was noticed on 2nd day of incubation, as in control set of petriplates, without any spice sample.
DISCUSSION
Functional properties of aromatic plant/spices etc. are contained in their volatile aromatic secrections commonly known as essential oils8 . A. sativum bulbs have 0.1%-0.25% essential oil, which is composed of 60% diallyl disulphide (Allicin), 20% diallyl trisulphide, 6% allyl propyl disulphide and diallyl sulphide9 . The inhibitory activity of essential oil of A. sativum is widely attributed to diallyl disulphide (Allicin). Allicin is highly volatile and is formed by the action of enzyme allinase on alliin (an odourless precursor), when fresh cloves/bulbs of A. sativum are cut or bruised10 . The mode of action of allicin to inhbit growth of bacterial strains is not yet well understood however, it may involve : hydrophobic and hydrogen bonding of active components of essential oil to membrane proteins, perturbation of membrane permeability, leakage of ions and other cell contents, inhibition of membrane embedded enzymes, destruction of electrons transport systems, disruption of proton motive force (PMF) and coagulation of cell contents leading to death. The ineffectivity of aqueous extract and powderd form of A. sativum bulbs towards test microbes in the present experiment may be attributed to the loss of allicin during drying of cloves/bulbs under ambient conditions. The greater susceptibility of g+ve bacterial strains towards crude juice and essential oil of A. sativum may be due to the absence of an outer membrane in their cell membrane which makes them more sensitive to external environmental changes such as temperature, pH , natural extracts, essential oils and other antimicrobial substances. On the other hand, the lipopolysacharides in the cell membrane of g-ve bacteria could provide a barrier to many antimicrobial agents, rendering these bacteria more resistant to certain agents than g+ve bacteria. It is worth mentioning here that crude extract more effectively arrested microbes during broth dilution technique than zone inhibition assay. This may be attributed to the direct contact of the microbes with liquid media which might have allowed the easy and quick diffusion of antimicrobial components of crude juice to the target site.
Conclusion
Present in vitro study indicate that essential oil of A. sativum inhibited food borne pathogens most effectively and may be considered for food preservation. Further studies should be undertaken to elucidate the safety, stability and organoleptic aspects of essential oil and its precise mode of action. Interactions of essential oil components with different food matrices during various food processing treatments must be the focal area of research before their commercialization as „biopreservatives?.
References:
1. Weber ND, Anderson DO, North JA, Murray BK, Lawson LD, Hughes BG. In vitro virucidal activity of Allium sativum (garlic) extract and compounds. Planta Med 1992; 58 : 417–23.
2. Shoji S, Furuishi K, Yanase R, Miyazaka T, Kino M. Allyl compounds selectively killed human deficiency virus-type 1-infected cells. Biochem Biophys Res Commun 1993;194 : 610–21.
3. Lun ZR, Burri C, Menzinger M, Kaminsky R. Antiparasitic activity of diallyl trisulfide (Dasuansu) on human and animal pathogenic protozoa (Trypanosoma sp., Entamoeba histolyica and Giardia lamblia) in vitro. Ann Soc Belg Med Trop 1994; 74:51–9.
4. Reuter HD, Koch HP, Lawson LD. 1996. Therapeutic effects and applications of garlic and its preparations. In: Koch HP, Lawson LD (eds) Garlic: the science and therapeutic application of Allium sativum L. and related species. Williams and Wilkins, Baltimore, pp 135–213.
5. Iroegbu CU, Nkere. Evaluation of the antibacterial properties of Picralima nitida stembark extracts. International J Mol Med Adv Sci 2005; 1: 182-9.
6. Kim HO, Park SW and Park HD. Inactivation of Escherichia coli 0157:H7 by cinnamic aldehyde purified from Cinnamomum cassia shoot. J Food Micro 2004; 21:105-10.
7. Azzouz MA, Bullerman LB. Comparative antimycotic effects of selected herbs,spices, plant components and commercial antifungal agents. J Food Prot 1982; 45: 1298-1301.
8. Pruthi JS. Spices and Condiments. National Book Trust, New Delhi, India, 1976; pp. 117–21.
9. Stoll V, Seebeck E. Allium compounds. I. Alliin the true mother compound of garlic oil. Helv Chem Acta 1948 ; 31:189.
10. Lawson LD. The composition and chemistry of garlic cloves and processed garlic. In: Koch HP, Lawson LD (eds) Garlic: the science and therapeutic application of Allium sativum L. and related species. Williams and Wilkins, Baltimore, 1996 ; pp 37–107.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License