IJCRR - 8(14), July, 2016
Pages: 33-40
Date of Publication: 21-Jul-2016
Print Article
Download XML Download PDF
ANTI-OXIDANTANT AND PHYTOCHEMICAL SCREENING OF ACALYPHA COMMUNIS (m\?ll.arg.) BY HPTLC FINGER PRINTING METHOD
Author: Rajasekaran S., R. Anandan
Category: Healthcare
Abstract:Aim: The aim of this current investigation is to evaluate the in-vitro antioxidant by various antioxidant assays and also the study, an attempt has been made to develop simple, precise and accurate HPTLC method
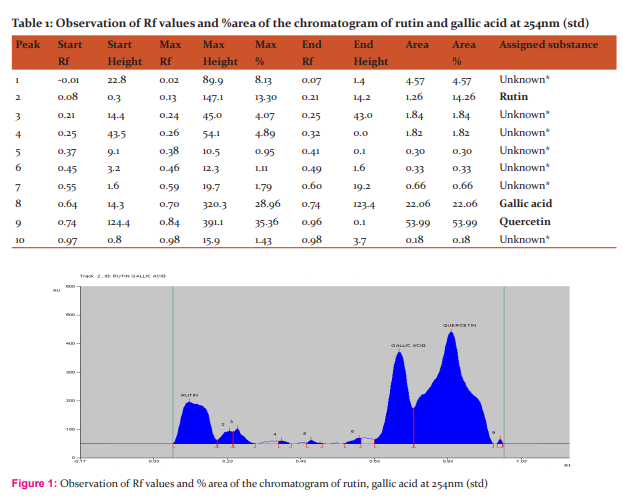
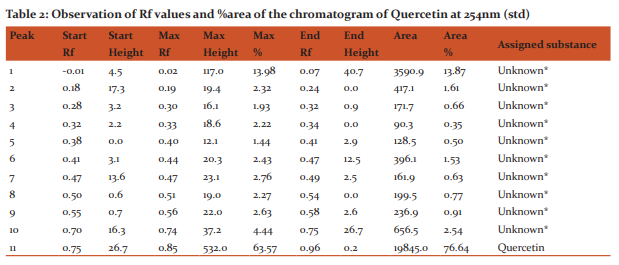
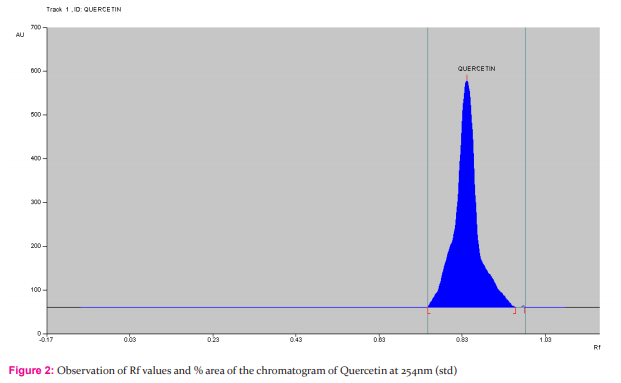
Materials and Methods: to evaluate the in-vitro antioxidant by various antioxidant assays such as DPPH scavenging, nitric oxide scavenging, and also the study, an attempt has been made to develop simple, precise and accurate HPTLC method by using Rutin, Gallic acid and quercetin as a standard marker compound with mobile phase of Toluene-Ethyl acetate-Formic acid-Methanol(3:6:1.6:0.4). The detection of Rutin, Gallic acid and quercetin were performed at 254 nm respectively.
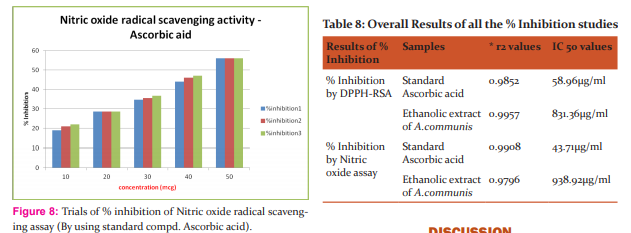
Results: The ability to scavenge the free radical, DPPH was measured at an absorbance of 517 nm. So the DPPH % inhibition of ethanolic extract of A.Communis plant showed that IC50 values 831.36\?g/ml (r2=0.9957) and Nitric oxide scavenging assay showed that IC50 values938.92\?g/ml and 805.85\?g/ml (r2=0.9766) respectively as compared to the standard of Ascorbic acid of 43..71\?g/ml (r2=0.9908) The detection of Rutin, Gallic acid and quercetin were performed at 254 nm respectively.
Conclusion: From the present work we conclude that species of A.Communis are highly potential in biological activity. The preliminary screening of the samples revealed the presences of high value class of compound like phenolic group as the major content in the plants. In terms of the Antioxident activity of A.Communis possess higher activity
Keywords: Acalypha communis, DPPH, Nitric oxide, HPTLC, Rutin, Quercetin
Full Text:
INTRODUCTION
India has a rich culture of medicinal herbs and spices, which includes about more than 2000 species and has a vast geographical area with high potential abilities for Ayurvedic, Unani, Siddha traditional medicines but only very few have been studied chemically and pharmacologically for their potential medicinal value [1,2]. Hence, natural products from medicinal plants need to be investigated by scientific methods for their anti-oxidant activity. The plant Acalypha communis is a synonym of Ricinocarpus communis (Müll.Arg.) belonging to the family of Euphorbiaceae. It includes herbs, shrubs and small trees, Shrubs or suffrutex frequently with resinous bright droplets on leaves and inflorescences; indumentum of simple or glandular hairs. Inflorescences spicate, usually unisexual [3].
MATERIALS AND METHODS
Collection, Identification and Authentification of the Plants. The leaves of Acalypha communis Müll. Arg., were collected from the malappuram district, Kerala, India, during the month of October 2013. The plant materials were identified and authenticated by Dr. Pradeep Botanist Calicut University, Kozhikode, Kerala. Voucher specimens were kept in our laboratory for future reference (437527AC). Preparation of extracts The granulated dried leaves of Acalypha communis (500 g) was packed in a Soxhlet apparatus and subjected to continuous hot percolation for 8 hrs using 450 ml of ethanol (95 % v/v) as solvent. The extract was concentrated to dryness under reduced pressure and temperature and dried in a desiccators (yield 75 g, 15 % w/w). The extract was suspended in 5 % gum acacia and used for further experiments. Preliminary phytochemical screening The extract were screened qualitatively for the presence of various groups of phytoconstituents using different chemical tests [4].
HPTLC STUDIES FOR QUALITATIVE ANALYSIS
Instrumentation In the present work Camag HPTLC system equipped with Linomat 5 applicator, twin trough chamber (20x10cm, 0.2 mm thick) size, TLC scanner 3, Reprostar 3 with 12bit CCD camera for photo documentation, controlled by WinCATS? 4 software were used. All the solvents used were of HPTLC grade obtained from Merck. All weighing were done on Precisa XB 12A digital balance. Procedure The Ethanolic extract (sample A) solutions were prepared. The TLC plate was heated at 1200C for 30 min prior to use. Ethanolic extract solution (2 μl), standard solutions of Rutin, Gallic acid and quercetin (0.2µg/μl) were applied in duplicate, as tracks 8, with a band length of 6.0 mm each on a precoated silica gel 60 F254 TLC plate, with Linomat V applicator using a Hamilton syringe (100µl).Mobile phase for ethanolic extract was toluene: ethyl acetate: formic acid: methanol (3:6:1.6:0.4). No prewashing of the plate was done. Chamber saturation time 5 minutes. The purpose of development TLC plate was kept for distance of 77 mm. Post derivatization has been done with vanillin?phosphoric acid. The derivatized plate was dried in hot air oven at 60 0 c for 5 minutes. and scanned at 254 nm, band length 6.0 mm, slit dimension 6.00x0.45mm,micro, scanning speed (20mm/s) and source of radiation was Deuterium and Tungsten lamps respectively. The Rf and peak area of the spots were interpreted by using software. The derivatized plate was photo documented under 254 nm light using Camag Reprostar 3, equipped with 12 bit CCD camera.
IN-VITRO ANTIOXIDANT POTENTIAL CAPACITY OF THE LEAVES OF ACALYPHA COMMUNIS: DPPH –
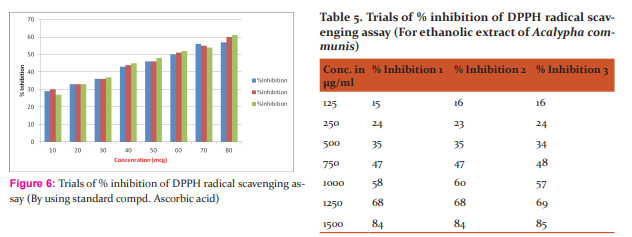
RSA method The free radical scavenging activity of ethanolic extract and the standard L-Ascorbic Acid (Vitamin C) was measured in terms of hydrogen donating or radical scavenging ability using the stable radical DPPH [5,6]. Here,0.1mM solution of DPPH in alcohol was prepared and it must be protected from light influence and 3ml of this solution was added to 1ml various concentration (125-1500 μg/ml) of extract or standard solution of (10-100 μg/ml). Absorbance was taken after 30 min at 517nm. Results are provided in Table (5,6) and Figure (6,7). The percentage inhibition activity was calculated from [(A0 -A1 )/A0 ] x 100, where A0 is the absorbance of the control and A1 is the absorbance of extract/standard taken as Ascorbic acid.
Nitric oxide scavenging assay
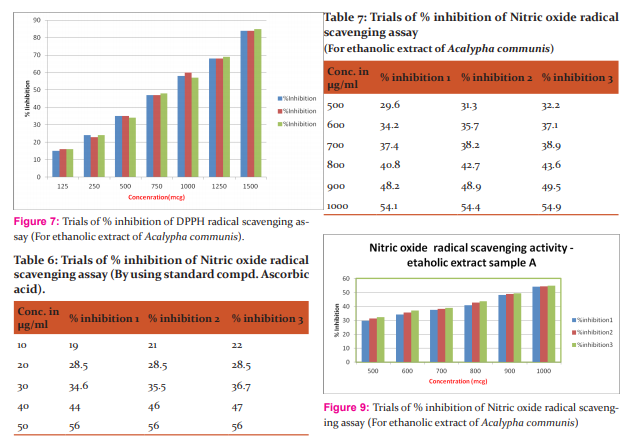
Nitric oxide radical inhibition was estimated by the use of Griess Illosvory reaction [7,8]. In this method, Griess Illosvory reagent was converted by using Napthyl ethylene diamine dihydrochloride (0.1%w/v) instead of the use of 1-napthylamine (5%). The reaction mixture (3ml) containing 2ml of 10 mM sodium 0.5ml saline phosphate buffer and 0.5ml of standard solution or ethanolic extracts of (500 -1000μg/ml) were incubated at 25°C for 150min. After incubation, 0.5ml of the reaction mixture was mixed with 1ml Sulfanilic acid reagent (0.33% in 20% glacial acetic acid) and allowed to stand for 5min for the completion of the reaction of diazotization. After that standard solution of dihydrochloride further 1ml added, mixed and was allowed to stand for 30min at 25°C. The concentration of nitrite was assayed at 546nm and was calculated with the control absorbance of the standard nitrite solution (without extract or standards, but the same condition should be followed). Here the blank is taken as the buffer and make up solvents and the Ascorbic acid (10 -50μg/ml) was taken as standard. Results are provided in Table (7,8 ) and fgure (8,9) The percentage inhibition was calculated using the formula:

Statistical analysis
The results of various studies were expressed as mean ± SEM and analyzed statistically using one way ANOVA followed by Dunnets Test to find out the level of significance. Data were considered statistically significant at minimum level of p < 0.05.
RESULTS
Preliminary phytochemical screening The preliminary phytochemical analysis of fractions of Acalypha communis shows presence of steroids, alkaloids, flavonoids, glycosides, saponins, tannin and carbohydrate.
HPTLC finger printing of Acalypha communis (sample A) The ethanolic extracts of Acalypha communis were subjected to generate HPTLC finger printing profile represented as chromatogram. The solvent system used in the investigation was found to give compact spots for extracts at different Rf values table no. 3
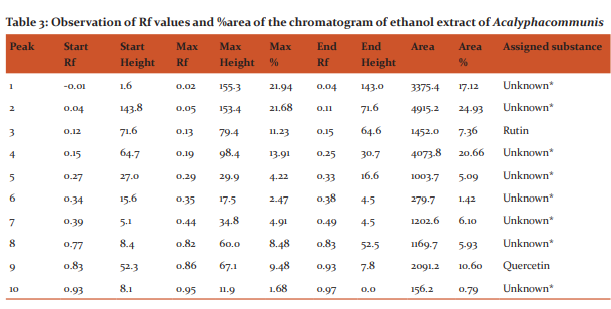
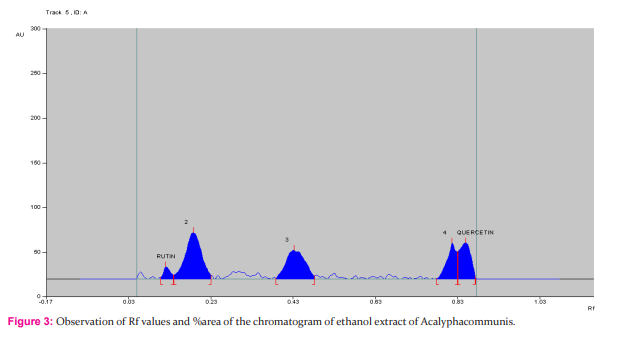
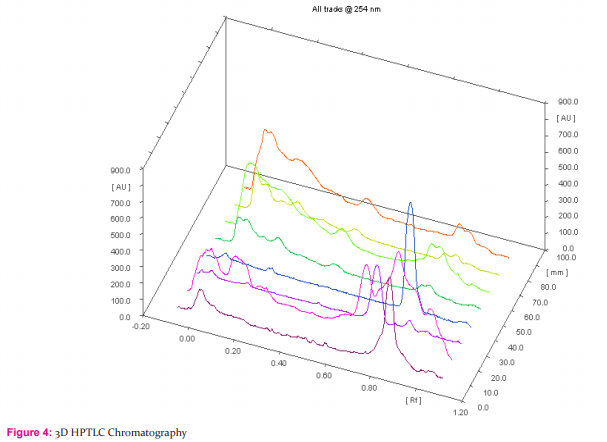

DISCUSSION
Preliminary phytochemical screening of the plants revealed the major class of compounds present in ethanolic extract of A.Communis like Alkaloids, Phenolic Compounds, Tannins, Steroids and Glycosides in major amounts.
Capacity of DPPH –RSA The antioxidant reacts with stable free radical, DPPH and converts it to 1, 1- Diphenyl-2- Picryl Hydrazine. The ability to scavenge the free radical, DPPH was measured at an absorbance of 517 nm. So the DPPH % inhibition of ethanolic extract of A.Communis plant showed that IC50 values 831.36μg/ml (r2=0.9957). Ascorbic acid has taken as reference which showed 58.96μg/ml. (r2=0.9852) among these results ethanolic extract of A.communis has more potent than traditionally claiming decoction. The results of % inhibition as shown in the Table:9 respective to IC50 values and regression r2 is the mean value of (n=3). The A.Communis leaves possesses the antioxidant activity and its substances may be potential responsible for the treatment of jaundice. So there are many scopes are there in leaves portion of this plant. Capacity of Nitric oxide scavenging assay Nitric oxide is a very unstable species under the aerobic condition. It reacts with O2 to produce the stable product nitrates and nitrite through intermediates through NO2 , N2 O4 and N3 O4 . It is estimated by using the Griess reagent. In the presence of test compound, which is a scavenger, the amount of nitrous acid will decrease. The extent of decrease will reflect the extent of scavenging, the %inhibition of ethanolic extracts of three parallel readings of (r2=0.9796) showed that IC50 values938.92μg/ml and 805.85μg/ml (r2=0.9766) respectively as compared to the standard of Ascorbic acid of 43..71μg/ml (r2=0.9908).
CONCLUSION
From the present work we conclude that species of A.Communis are highly potential in biological activity. The preliminary screening of the samples revealed the presences of high value class of compound like phenolic group as the major content in the plants. In terms of the Antioxident activity A.Communis possess higher activity.
References:
1. Sandhu DS, Heinrich M (2005). The use of health foods, spices and other botanicals in the sikh community in london. Phytotherapy res.19:633-42.)
2. Gupta, M.P., Solis P.N., Calderon, A.I., Guionneau Sinclair, F., Correa, M., Galdames, C. Guerra, Espinosa, A., Alvenda, G.I., Robles, G., Ocampo, R (2005) Medicinal ethnobotany of the teribes of bocasdel toro, panama. J Ethnopharmacol, 96, 389-401.
3. Hayden, W.J. and Hayden, S.M. 2000. Wood anatomy of Acalyphoideae (Euphorbiaceae). I. A. W. A. Bull. 21: 213–235.
4. Kokate CK. In: Practical Pharmacognosy, Preliminary Phytochemical Screening, first ed., Vallabh Prakashan, New Delhi, 1986; 111.
5. Havsteen B. Flavonoids, a class of natural products of high pharmacological potency. Biochem Pharmacol. 1983;32:1141–1148.
6. Mohammad T.A., Determination of Polyphenolic content and free radicalscavenging activity of Flemingia strobilifera, Advances in Pharmacology and Toxicology.2009;10 (1):89-95.
7. Chakraborthy G.S.,Free radical scavenging activity of Costus speciosusleaves, Indian Journal of Pharmaceutical Education and Research.2009;43(1):96-98.
8. Hyoung Lee S., Antioxidant activity of browing reaction products isolated from storage aged orange juice, J agric Food Chem1992.40(4): 550-552.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License