IJCRR - 3(10), October, 2011
Pages: 75-82
Print Article
Download XML Download PDF
SWELLING STUDIES OF Gt-cl-poly(AA) WITH RESPONSE TO TIME,TEMPERATURE AND pH PROPERTIES
Author: Prashar Deepak
Category: Healthcare
Abstract:Cross-linked three dimensional network of Gum tragacanth with acrylic acid has been synthesized using
ascorbic acid-potassium persulphate as an initiator and glutaraldehyde as a crosslinker. The polymer
formed was further studied for swelling behavior in response to time, temperature and pH.
Keywords: crosslinked, Gum tragacanth, swelling studies, pH
Full Text:
INTRODUCTION
Superabsorbent polymers (SAP) are the polymers that can absorb and retain extremely large amounts of a liquid relative to their own mass [1]. Water absorbing polymers, which are classified as hydrogels, when cross-linked [2] absorb aqueous solutions through hydrogen bonding with water molecules. Hydrogels are hydrophilic, three-dimensional networks, which are able to imbibe large amount of water or biological fluids. Hydrogel formation is also known as the complexation of the polymers [3, 4]. Their ability to swell under biological conditions makes it an ideal material for use in drug delivery [5, 6] and immobilization of proteins, peptides and other therapeutically active compounds [7]. Swelling characteristics of the hydrogels are affected by the properties of the swelling medium such as pH [8], ionic strength [9] and counter ions [10]. Many researchers have studied the synthesis, characterization, swelling behavior and controlled drug delivery of the polymer hydrogels [11, 12]. These days modified polysaccharides have been the major area of scientific research. Lot of work has been carriedout on various polysaccharides such as starch, cellulose, chitosan, dextrin, guar-gum, psyllium and many more. The Gum tragacanth is a complex mixture of polysaccharides and is composed of two major components: 60-70% water insoluble fraction called bassorin and 30% soluble fraction of tragacanthin. Water-soluble tragacanthin is a branched arabiogalactan, which is soluble in 70% ethanol. The acidic bassorin has a chain of (1-4)-linked α-D-galacturonic acid units some of which are substituted at O-3 with β-Dxylopyranosyl units and some of these being terminated with galactose or fucose. Tragacanth is stable over a wide pH range as low as pH 2. Gum tragacanth is a highly branched, heterogenous hydrophilic carbohydrate polymer, which may contain methoxyl groups. It is complex, slightly acidic polysaccharide bounded with small proportions of proteins and with trace amounts of starch and cellulosic materials. Ca Mg and K are also present as associated cations. On acid hydrolysis, Gum tragacanth produces sugars of D-galacturonic acid, D-galactose, Lfucose (6-deoxy-L-galactose), D-xylose, Larabinose and L-rhamnose. D-galacturonic acids repeating units forms the main chain by 1, 4- linkages and a short side chains consisting of Dxylose residue connected to the main chain by 1, 3-linkages. Further this D-xylose residue is attached to L-fucose and D-galactose by 1, 2- linkages. L-fucose may be replaced by Dgalactose. In the structure of tragacanthin, several chains are arranged in parallel. In present paper, a cross-linked three dimensional network of Gum tragacanth with acrylic acid has been synthesized using ascorbic acid-potassium persulphate as an initiator and glutaraldehyde as a crosslinker. The polymer formed was further studied for swelling behavior in response to time, temperature and pH
MATERIALS AND METHOD
Gum tragacanth (sd Fine Chemicals Pvt. Ltd.) and ascorbic acid-potassium persulphate (sd Fine Chemicals Pvt. Ltd.) were used as backbone and initiator, respectively. Glutaraldehyde (MERCK) and acrylic acid (MERCK) were used as crosslinker and monomer, respectively. Instrumental Analysis Scanning Electron Micrographs (SEM) of the candidate polymers were taken on LEO-435VF, LEO Electron Microscopy Ltd
Synthesis
1.0 g of Gum Tragacanth was taken in a reaction flask containing 25ml of distilled water. To the reaction mixture 0.5mol L-1 of Acrylic acid was added followed by the addition of Ascorbic acidKPS in 1:1.25 molar ratios as an initiator system and 0.42mol L-1 of glutaraldehyde as a crosslinker. The reaction was carried out at pH 7.0 for 90min at 40OC. At the end of the reaction the homopolymer was removed on washing with hot water and the gel obtained was allowed to stand for about 10-12 hours undisturbed for gelling process to take place. The product obtained was dried in the oven at 60 OC till a constant weight was obtained. The percentage grafting and percentage swelling was calculated as per the following equations 1 and 2:

Preparation of solutions of different pH
Alkaline solutions were prepared with NaOH pallets and acidic solutions were prepared by the addition of HCl and pH was determined with Cyberscan 1100, EUTECH INSTRUMENTS, pH meter.
Preparation of artificial biological fluid
Artificial biological fluid was prepared in the laboratory using electrolytic powder and distilled water (DW). 25gms of electrolytic powder was dissolved in per 1000ml of distilled water to obtain the artificial biological fluid of 25000ppm.
RESULTS AND DISCUSSION
Mechanism
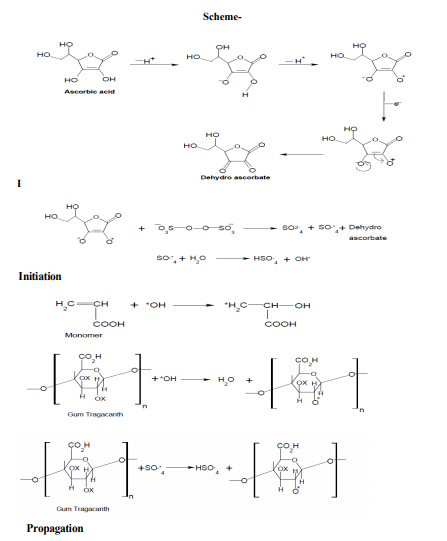
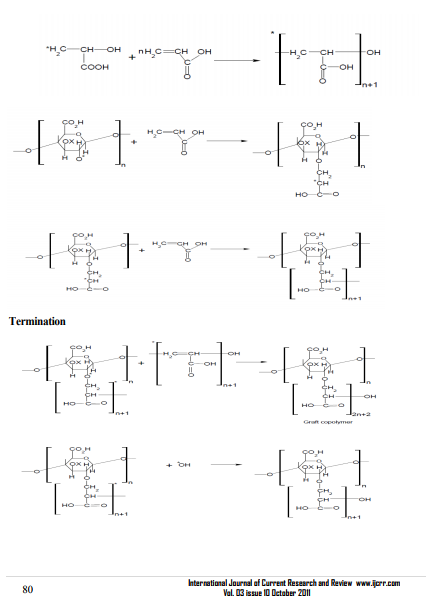
Hydroxyl groups present on the backbone and monomers are the active sites for graft copolymerization to take place. Various steps involved in the graft copolymerization of Gum tragacanth with different vinyl monomer mixtures are depicted in Scheme-I: Initially, ascorbic acid ion reacts with the potassium persulphate to generate SO4 -* which on further reaction with water molecule generates OH* followed by the interaction of OH* and SO4 -* with backbone and monomer resulting in generation of active sites. Activated monomer and backbone molecules propagate further and give rise to three dimensional crosslinked networks in the presence of glutaraldehyde. However, chain termination takes place either by the reaction of OH* with the live propagating macromolecular chains or reaction between two activated chains.
Characterization
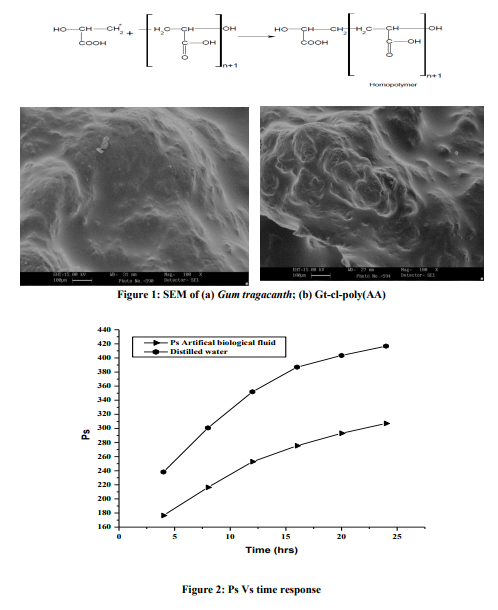
SEM
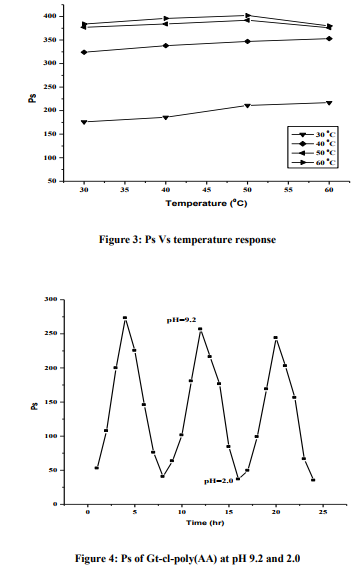
n order to have the conducting impact, the samples were gold plated and the scanning was synchronized with microscopic beam so as to maintain the small size over a large distance relative to the specimen. A remarkable three dimensional appearance with high resolution was obtained in case of crosslinked product. SEM images clearly exhibited the differences in the surface morphology of the Gum Tragacanth and Gt-cl-poly(AA) which were brought about through copolymerization and crosslinking (Figures. 1a-b). Water uptake studies of Gt-cl-poly(AA) Effect of time Water uptake studies were carried out at different time intervals (4, 8, 12, 16, 20 and 24hrs) using distilled water and artificial electrolytic biological fluid. It was found that with increase in time, Ps increases but the rate of swelling slowed down after 20 hrs and attains a constant value at 24 hrs (Figure 2). Moreover, the swelling capacity of candidate polymer in artificial biological electrolytic fluid is lower in comparison to distilled water, which could be explained on the basis of reverse osmosis process. In case of electrolytic fluid and in hypertonic solutions (higher concentration) the polymer showed deswelling behavior. Similarly after 24 hrs, the pores of the network structure got saturated with the solvent molecule there by leaving no more space for further solvent entry into the matrix. Effect of temperature Water uptake studies opting the different temperature (30, 40, 50, 600C) were carried out. Ps increase with increase in temperature upto 500C and decrease thereafter (Figure 3). This might be due to the reason that at a higher temperature, the polymer becomes compact leading to desorption. Effect of pH Pulsatile behavior of bio-polymer was analyzed in solutions of different pH and percentage swelling was observed to be significantly affected (Figure 4). When the completely dried hydrogel sample is placed in the swelling medium of pH 9.2, the solvent diffuses into the outer surface of the candidate polymer through the micropores, resulting in the plasticization of macromolecular chains. At the same time, the carboxylic groups, attached along the polymer backbone, undergo ionization to yield –COO– groups (since the pH of the swelling medium is more than the pKa value of acrylic acid inside the gel matrix). This results in the formation of a charged hydrated layer through which the counter ions along with the solvent molecules embed into the interior dry core region and allow the gel to swell. In this way, the dry core slowly disappears and the gel matrix continuous
to swell. The swelling is further enhanced due to relaxation (or unfolding) of macromolecular chains owing to the repulsion among similarly charged –COO– groups which also promotes the swelling process. When the fully hydrated gel is placed in the medium of pH 2.0, H+ ions present in the external solution, diffuse into the gel matrix through water-filled macrospores that have existed in the fully hydrated gel. These H+ ions protonate the –COO– groups to yield uncharged –COOH groups which ultimately results in folding of the macromolecular chains as the repulsive forces no longer exist, thus letting the solvent molecules to come out of the polymer matrix resulting in deswelling.
CONCLUSION
Modification of Gum tragacanth via crosslinking and network formation with acrylic acid improves the property profile and usability of the polymer in various technical fields. Further, polymer was found to show pulsatile behavior under acidic and basic conditions, which could be of great significance in the sustained release of colon specific drugs.
 \
\
References:
REFERENCES
1. Horie K, Baron M, Fox RB, He J, Hess M, Kahovec J et al. Definitions of terms relating to reactions of polymers and to functional polymeric materials (IUPAC Recommendations 2003). Pure Appl. Chem. 2004; 76 (4): 889.
2. Kabiri K. Synthesis of fast-swelling superabsorbent hydrogels: effect of crosslinker type and concentration on porosity and absorption rate. Eur. Polym. J. 2003; 39: 1341.
3. Peppas NA, Khare AR. Preparation, structure and diffusional behavior of hydrogels in controlled release, Adv. Drug Deliv. Rev. 1993; 1: 35.
4. Lee SH, Shin H. Matrices and scaffolds for delivery of bioactive molecules in bone and cartilage tissue engineering, Adv. Drug Deliv. Rev. 2007; 59(4-5): 339.
5. Wakerly Z, Fell JT, Attwood D, Parkins, DA. Pectin/ethylcellulose film coating formulations- For colonic drug delivery. J. Pharm. Pharmacol. 1997; 49: 1210.
6. Ashford M, Fell J, Attwood D, Sharma H, Woodhead P. Studies on pectin formulations for colonic drug-delivery, J. Control. Rel. 1994; 30: 225.
7. Chen X, Martin BD, Neubauer T K, Linhardt RJ, Dordick JS, Rethwich DG. Enzymatic and chemo enzymatic approaches to synthesis of sugar-based polymer and hydrogels. Carbohydr. Polym. 1995; 28(1): 15.
8. Casadei MA, Pitarresi G, Calabrese R, Paolicelli P, Giammona G. Biodegradable and pH-sensitive hydrogels for potential colon-specific drug delivery: characterization and in vitro release studies. Biomacromol. 2008; 9, 43.
9. Chiu HC, Wu AT, Lin YF. Synthesis and characterization of acrylic acid-containing dextran hydrogels. Polym. 2001; 42, 1471.
10. Gils PS, Ray D, Mohanta GP, Manavalan R, Sahoo PK. Designing of new acrylic based macroporous superabsorbent polymer hydrogel and its suitability for drug delivery. Int. J. Pharm. Pharmaceu. Sci. 2009; 1: 44.
11. Kaith BS, Jindal R, Mittal H. Superabsorbent hydrogels from poly(acrylamide-co-acrylonitrile) grafted Gum ghatti with salt, pH and temperature responsive properties Der Chemica Sinica 2010; 1 (2), 92.
12. Ganji F, Farahani EV. Hydrogels in Controlled Drug Delivery Systems. Iran. Polym. J. 2009; 18 (1): 63.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License