IJCRR - 8(17), September, 2016
Pages: 13-21
Date of Publication: 11-Sep-2016
Print Article
Download XML Download PDF
ISOLATION AND CHARACTERISATION OF DIATOMS FROM WATERBODIES OF MUMBAI
Author: Disha Rana, Deepika Bhandari
Category: Healthcare
Abstract:Diatom plays an important role in Forensic Science and in the studies of water quality. Diatom acts as a supportive evidence for ascertaining the cause of death as well as the place of drowning. Presence of diatoms in the distant vital organs is an important method to distinguish between ante-mortem and post-mortem drowning. It is possible to pinpoint the site of drowning, if diatom species recovered from the tissues of the corpse are compared to that found in the water body from where the corpse is recovered. In the present study, 10 water bodies located in various parts of Mumbai were sampled for the study. Isolation of diatoms was carried out using acid digestion method. Total 35 species of diatoms were isolated and characterised. The diatom species identified belong to the following genera: Gyrosigma, Navicula, Melosira, Pinnularia, Surirella, Hantzschia, Eunotia, Fragilaria, Nitzschia, Gomphonema, Cymbella, Cyclotella, Neidim, Caloneis, Synedra, Epithemia, Frustulia. The information obtained helped to create a reference database on diatoms which can be utilized in future for ecological assessment and ready reference in drowning cases
Keywords: Diatoms, Drowning, Genera and species from Mumbai water bodies, Acid digestion method
Full Text:
INTRODUCTION
Diatoms are the most common types of phyto-planktons in aquatic ecosystems. They have unique feature called frustules. The frustules are cell walls composed of two valves, one overlapping the other and are made up of silica (SiO2 ). It is through frustules that different varieties of diatoms can be distinguished. By adding concentrated nitric acid (HNO3 ) to suspected materials, diatoms can be easily distinguished from other algal group. The acid will have no effect or will cause no injury to the diatoms as their silica cell walls do not decompose. Particularly in India, diatoms as compared to other group of algae are less studied due to several reasons such as scarcity of literature, tedious process of isolation, eye straining study/ observation as they are visualized under oil immersion. The diatoms may fall under one of the following three classes:
1. Class: Coscinodiscophyceae Order: centrales or centric diatoms with symmetry about a point (radial)
2. Class: Fragilariophyceae Order: pennales or pennate diatoms with symmetry about a line (bilateral) and without a raphe (araphids)
3. Class: Bacillariophyceae Order: pennales with a raphe (raphids) Professor H. Gandhi has added much to our knowledge in the field of diatomology/ limnology and is considered as the father of limnology in India. In India, the early records of diatoms are those of Ehrenberg (1854), Dickie (1882), Carter (1926), Majeed (1935), Skvortzow (1935) and Biwas (1936), Gangla (1949), Venkataraman (1939,1956), Krishnamurthy (1954), Gandhi (1952), Desikachary (1954), Lakshminarayana (1962), Thomas (1965), Zahamensk (1973), Pandey and Pandey (1980), Somashekar (1983), Sarode and Kamat (1984),Mann and Droop (1996),Tarar and Bodhke (1998),Kociolek and Spaulding (2000), Kociolek and Stoermer (2001), Bhagat (2002), Mishra and Mishra (2002), Kilroy et al. (2003), Mishra (2006), Patil and Kumawat (2007), Vanormelingen et al.(2008), etc. The first to record the diatom from Maharashtra was probably Gonzalves(1947).
MATERIALS AND METHODS
Collection of Water Samples: The water samples was collected during the month of February from 10 selected water bodies of Mumbai, India. The samples were collected in different reagent bottles. Before the samples were collected in the bottle, the floor of the water bodies was disturbed so that more amount of mud enters the bottle, thereby increases the number of diatoms in the samples collected. The bottles were labelled mentioning the place, date, time of collection and sample number. The mud was allowed to settle down in the bottle. Digestion Method: Nitric acid digestion method was used. The water sample (approximately 1 ml) from the bottom of the bottle was collected using dropper and was transferred in vials of 1.5 ml .The sample was then digested by adding 4 drops of concentrated nitric acid and incubated overnight at room temperature. Cleaning of Diatoms: The samples were centrifuged at 7000 rpm for 7 minutes. The supernatant was decanted and replaced with distilled water. The sample was made transparent by vortexing it at 7000 rpm for 5 minutes. This process of centrifuging and vortexing was repeated thrice to produce a pellet. The supernatant was decanted and the pellet was used for further examination. Preservation of Diatoms: The cleaned diatoms were preserved in 4% formalin added with glycerine. Mounting: The sediments were taken on slides and examined under compound microscope (Olympus CX41; Model: CX41RF) (magnification up to 100X)
RESULTS
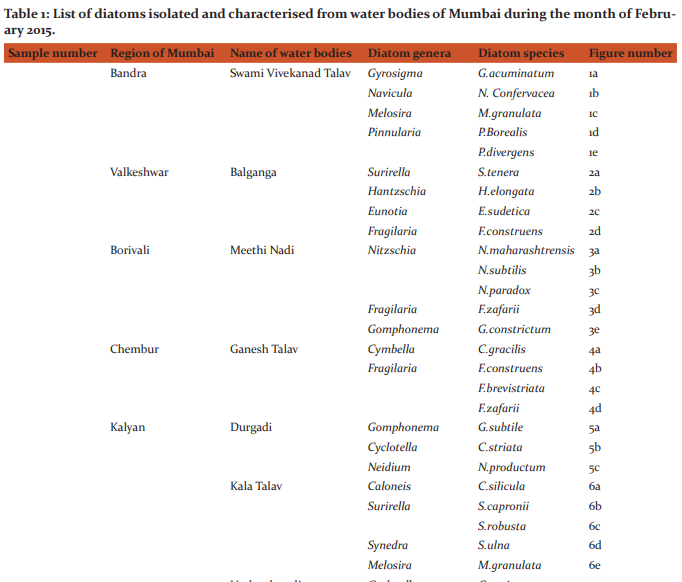
Table 1 represents the list of diatoms isolated and characterised from different water bodies of Mumbai. Description of various species found during study period is as follows: (Figures 1, 2, 3, 4, 5, 6, 7, 8, 9 and 10). Gyrosigma acuminatum(Kuetz.) Rabh. Frustules solitary; valves 99-106.5μ long, 11.9-12.8μ broad, sigmoid, lanceolate in outline, gradually tapering from the middle towards the ends; ends broadly rounded; raphe sigmoid and central; axial area narrow; central area small and elliptical; striae 18 in 10μ, transverse and longitudinal striae at equal distances from one another. (Figure 1a) Navicula confervacea Kuetz.f.nipponica Skv. Frustules broadly rectangular in girdle view, united together to form filaments; valves 10-15μ long, 5-6.5μ broad, elliptic, with obtusely rounded ends; axial and central areas broad, lanceolate; striae 22-24 in 10μ, marginal and radial. (Figure 1b) Melosira granulata( Ehr .)Ralfs Frustules 5.5-8.3μ in diameter, short, cylindrical, united in short or long chains; semi-cells 7-25.6μ high; end cells with spines, furrows and straight rows of areoles; row of areoles 10-14 in 10μ, spirally disposed. (Figure 1c) Pinnularia borealis Ehr. Valves 30.2-42μ long, 7.5-9.2μ broad, linear, elliptical with broadly rounded ends; axial area somewhat widened in the middle; raphe thread like with strongly hooked terminal fissures; striae 5-6 in 10μ, coarse, slightly radial in the middle. (Figure 1d) Pinnularia divergens W.Smith. Valves 75-85μ long, 14-15μ broad, linear lanceolate with constricted, broadly produced rounded ends; raphe thin and somewhat undulate with central pores unilaterally bent and terminal fissures thick, bayonet shaped; axial area linear; central area large, rhomboid, reaching the margins with conical projections formed into the sides; striae 9-10 in 10μ, thick, strongly radial in the middle and convergent at the ends. (Figure 1e)
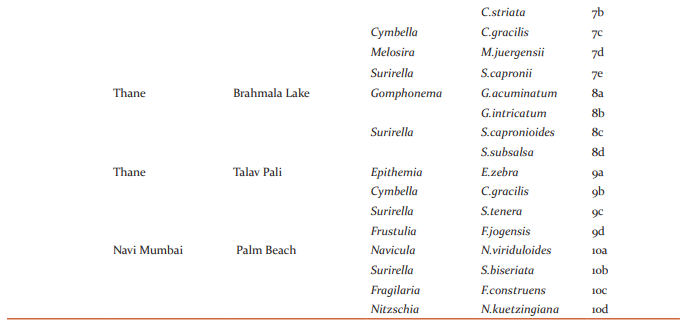
Surirella tenera Greg.v.ambigua Gandhi Valves 88-102.3μ long,22-26.2μ broad, robust, hetero-polar, linear ovate with rounded apex and cuneate rounded base; axial field narrowly lanceolate with a median line; marginal folds strongly developed with distinct projections; costae 18- 22 in 100μ, strong, radial at the ends; striae indistinct.(Figure 2a and 9c) Hantzschia elongate (Hantz.) Grun. Valves 10-239μ long,10.5-11.5μ broad, slender, linear with parallel sides, sometimes bent in the middle; ends constricted, narrowed and produced, slightly backwardly bent and sub-capitate; keel excentric, keel punctae 8-9 in 10μ,fine, and indistinctly punctate.(Figure 2b) Eunotia sudetica O.Muell. Valves 54-56.8μ long,5.5-7.2μ broad, linear, slightly arcuate; dorsal margin convex, ventral margin straight or slightly concave; ends slightly detached; end nodules centrally retracted, inner nodules well visible; striae 12-14 in 10μ,feebly radiating towards the ends.(figure 2c). Fragilaria construens (Ehr.)Grun.v.venter Grun.f.Pusilla Grun. Frustules linear, attached together to form short chains; valves 12-15μ long, 2.8-3μ broad, linear lanceolate, gradually tapering towards the ends; ends somewhat broadly rounded; pseudoraphe narrow, linear; central area not formed; striae 14-16 in 10μ,strong.(figure 2d,4b and 10c).
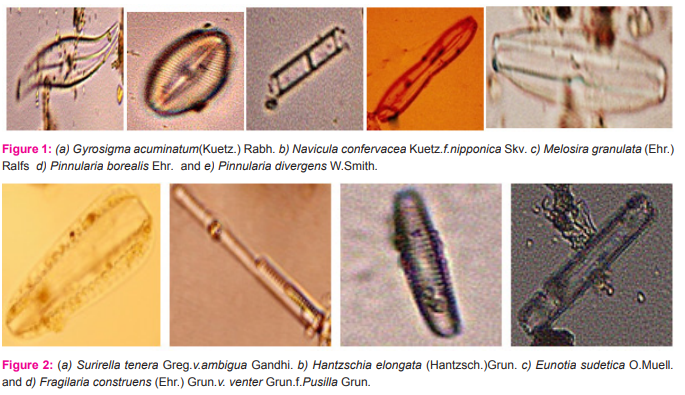
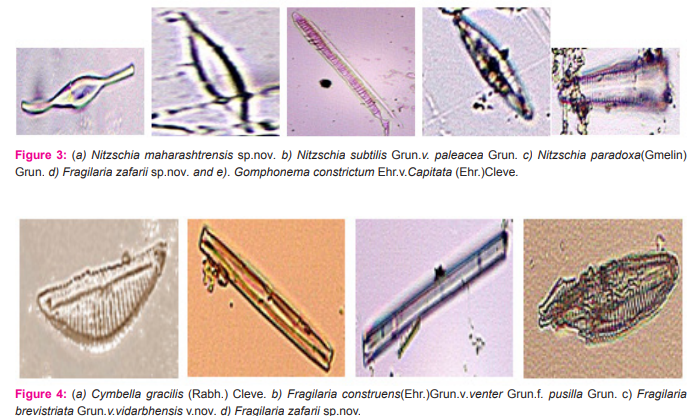
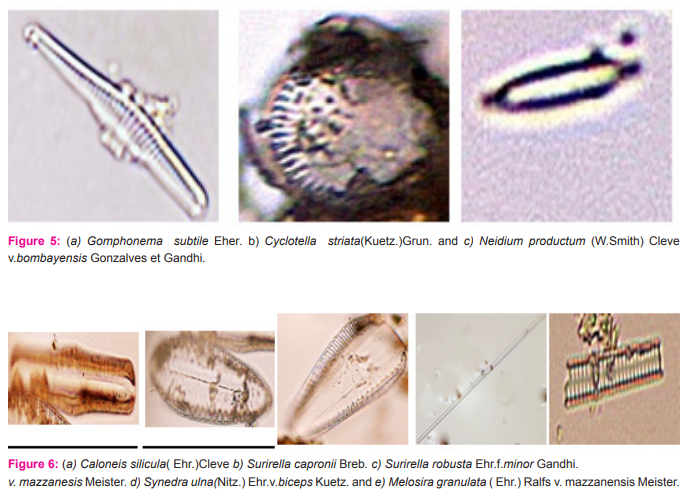
Nitzschia maharashtrensis sp.nov. Valves 45.5-63μ long,3.5-5.2μ broad, linear to linear lanceolate, tapering towards the ends, with prolonged, very slightly capitates ends; keel excentric, keel punctae 28-30 in 10μ,very small; striae not clearly visible.(Figure 3a) Nitzschia subtilis Grun.v. paleacea Grun. Valves 26-35μ long, 2.2-2.7μ broad, narrowly lanceolate with acute ends; keel punctae 16-18 in 10μ, small; striae more than 30 in 10μ, indistinct.(Figure 3b). Nitzschia paradoxa(Gmelin)Grun. Frustules linear, rectangular, united in bundles in girdle view; valves 91-121.5μ long,4.5-5.5μ broad, linear, spindle shaped; keel central, keel puntae 6- in 10μ,forming a row in the middle portion of the valve; striae about 30 in 10μ,very fine.(figure 3c). Fragilaria zafarii sp.nov. Frustules in short chains, rectangular in girdle view; valves 52.2μ long,7.5-10.5μ broad, linear lanceolate or rhomboid with gradually tapering somewhat acutely rounded ends; pseudo-raphe narrow; central area slightly bilateral with an elliptic margin enclosing two mucilage pores; striae 9-10 in 10μ,coarse.(figure 3d and 4d). Gomphonema constrictum Ehr.v.capitata (Ehr.)Cleve. Valves 55-63μ long,12-13.3μ broad, broadly clavate with feebly constricted, broadly rounded subtruncate apex and attenuated rounded base;raphe thick and straight; axial area narrow, linear; central area somewhat rhomboid, slightly unilateral with a stigma on the opposite side; striae 10-12 in 10μ,coarse and radial, alternately long and short in the middle.(figure 3e). Cymbella gracilis(Rabh.)Cleve. Valves 137-148μ long, 22-23μ broad, linear with broadly rounded ends; raphe thick, strongly complex and folded with central pores prominent and terminal fissures thick and obliquely comma shaped; axial area about ¼ the breadth of the valves, linear; central area slightly dilated; striae 5-6 in 10μ, thick, radial in the middle and convergent at the ends with fairly broad longitudinal bands.(figure 4a and 7c and 9b). Fragilaria brevistriata Grun.v.vidarbhensis v.nov. Frustules linear, loosely attached together to form short chains in girdle view; valves 19.7-23μ long,3.5-4.8μ broad, linear lanceolate, strongly tumid in the middle and slightly inflated towards the end with somewhat acutely rounded ends; pseudoraphe broad in the middle, linear lanceolate; striae 11-13 in 10μ,thick.(figure 4c). Gomphonema subtile Eher. Valves 32-45μ long, 5-6μ broad, narrowly lanceolate clavate, delicate, with slightly capitate, broadly rounded apex and gradually attenuated, produced rounded base; raphe thin and straight; axial area narrow; central area unilateral with an isolated stigma on the opposite side; atriae 12-14 in 10μ,distinctly punctuate.(figure 5a). Cyclotella striata(Kuetz.)Grun. Valves with strong wavy margins in the girdle view and more or less broad evenly striated border; valves discoid,14.5-25μ in diameter; central field with flexes and coarsely punctate; striae 7-9 in 10μ.(figure 5b and 7b). Neidium productum (W.Smith)Cleve v.bombayensis Gonzalves et Gandhi. Valves 85-103μ long,18-27μ broad, linear elliptical with slightly undulate margins and abruptly constricted produced, broadly rounded ends; raphe thin and straight with central pores bent in opposite directions and terminal fissures bifurcated; axial area wide; central area large and transversely elliptical; striae 20-22 in 10μ,radial,finely punctuate, crossed by longitudinal furrows near the margins.(figure 5c). Caloneis silicula( Ehr.)Cleve Valves 52-55μ long,8-9.9μ broad, linear, slightly swollen broadly rounded ends; raphe thin with distinct central pores and hooked terminal fissures; axial area narrow; central area large and elliptic, sometimes reaching the margins; striae 22- 24 in 10μ,fine,parallel but becoming radial and longer towards the ends, crossed by a fine longitudinal line near the margin.(figure 6a).
Surirella capronii Breb. Valves 13-183μ long,55-3μ broad, hetero-polar, ovate with broadly rounded, somewhat narrowed apex and acutely rounded base; middle line present, discontinuous, with strong spines developed on elevated cushions; axial area narrowly lanceolate; marginal folds strongly developed, with clear projections, flap windows quite evident;costae;9-13 in 100μ,thick,radial at the ends; striae fairly visible.(figure 6b and 7e). Surirella robusta Ehr.f.minor Gandhi. Valves 62-69.5μ long,30-35μ broad, hetero-polar, ovate with broad rounded ends; middle line absent; axial field linear lanceolate; marginal folds strongly developed; costae at unequal distances; striae indistinctly radiate.(figure 6c). Synedra ulna(Nitz.) Ehr.v.biceps Kuetz. Valves 298-455.2μ long, 5.9-7.1μ broad, linear, slightly bent, with swollen broadly subcapitate ends; pseudo-raphe narrow; striae 7-8 in 10μ,very coarse.(figure 6d). Melosira granulata( Ehr .)Ralfs.v.mazzanensis Meister. Frustules 12.2-15μ in diameter, short, cylindrical, flat, united in chains; semi-cells 8.5-11μ high; end cells with spines and furrows; rows of areoles 8-9 in 10μ .(figure 6e). Cyclotella antique W.Smith Valves 11-14.5μ in diameter, discold central field finely punctate and with 4-5 triangular depressions forming a ring; striae 18-20 in 10μ,punctate.(figure 7a). Melosira juergensii Agardh. Frustules 9.5-12μ in diameter, cylindrical, united in chains; outer mantal line straight, inner slightly wavy; neck absent; semi-cellls 13.5-15μ high; cell wall finely punctuate; rows of punctae 30 in 10μ,very faint.(figure 7d). Gomphonema acuminatum Ehr. v. turris Ehr. Valves 5-65μ long,12-14μ broad, clavate with gradually tapering lower half and biconstricted upper half with subcapitate apex and obtuse to subacute base; raphe somewhat thick; axial area narrow; central area small, unilateral with an isolated stigma on the opposite side; striae 10-12 in 10μ,slighty radial and distinctly punctate.(figure 8a). Gomphonema intricatum Kuetz.v.fossile Pant. Valves 95-115μ long,9.5-11.5μ broad, narrowly lanceolate, slightly inflated in the middle, with septate broadly rounded apex and gradually tapering rounded base; raphe somewhat thickened with distinct terminal fissures and unilateral central nodules; axial area broad; central area moderate with an isolated stigma on the opposite side and with two coarse punctae on one side of the central nodules; striae 8-9 in 10μ in the middle and 9-10 in 10μ towards the ends, radial, slightly curved and coarsely punctate.(figure 8b). Surirella capronioides Gandhi. Valves 80-130.6μ long,42-49 broad, hetero-polar, ovate with cuneate base; axial field narrowly lanceolate with a middle line interrupted throughout and with spines at both ends; flap marginal with clear flap projections; costae 24-26 in 100μ,strong,radial at the ends; striae fine and indistinct.(figure 8c). Surirella subsalsa W. Smith Valves 25-34.5μ long, 9.8-13μ broad, hetero-polar, ovate or ovate lanceolate with cuneate rounded base; axial field with projections; costae 32-40 in 100μ distinct (figure 8d). Epithemia zebra (Ehr.)Kuetz Frustules rectangular in girdle view; valves 16-113μ long, 8.5-10.5μ broad, arcuate with dorsal side convex and ventral side concave; ends very slightly or not constricted, narrow to obtusely rounded; raphe in the raphe canal, reaching 1/3-1/2 the breadth of the valve; costae 3-4 in 10μ, strong and radial, alternating with 3-5 rows of alveoli, rows of alveoli 12-13 in 10μ.(figure 9a). Frustulia jogensis Gandhi. Valves 56-59.6μ long,13-13.4μ broad, linear lanceolate with constricted, produced beak like rounded ends; raphe thin, delicate and straight, enclosed between the siliceous ribs; axial area very narrow, linear; central area small; striae about 32 in 10μ,perpendicular to the middle line, finely punctate, punctae arranged in straight longitudinal rows.(figure 9d). Navicula viriduloides Gandhi v.lanceolata Gandhi Valves 28-39.5μ long,.8-9μ broad, lanceolate with rostrate rounded ends; raphe thin, in between siliceous ribs with central pores unilaterally bent and terminal fissures curved; axial area narrow; central area suborbicular; striae 13-15 in 10μ,radial and curved in the middle and convergent at the ends.(figure 10a).
Surirella biseriata Breb.v.diminuta A.Cl Valves 1.5-25μ long,7.8-9.5μ broad, isopolar, linear lanceolate or elliptic lanceolate with acutely rounded ends; axial area linear to linear lanceolate; middle line indistinct; flap margin distinct; costae 30-35 in 100μ,quite evident and radial.(figure 10b).
Nitzschia kuetzingiana Hilse
Valves 26.2-32μ long,5.5-6μ broad, lanceolate with strongly produced constricted, small capitates ends; keel punctae 14- 16 in 10μ,small striae more than 30 in 10μ,fine and almost indistinct.(figure 10d).


DISCUSSION
In the present study, 35 diatom species under 17 genera were identified from 10 waterbodies of Mumbai, Maharashtra, India. The selected water bodies are as follows: Bandra(Swami Vivekanadare Talav), Valkeshwar (Balganga), Borivali (Meethi Nadi), Chembur(Ganesh Talav), Kalyan(Durgadi, Kala Talav, Umberdenadi), Thane (Brahmala Lake, Talav Pali) and Navi Mumbai(Palm Beach). The diatoms species identified belong to the following genera: Gyrosigma, Navicula, Melosira, Pinnularia, Surirella, Hantzschia, Eunotia, Fragilaria, Nitzschia, Gomphonema, Cymbella, Cyclotella, Neidium, Caloneis, Synedra, Epithemia, Frustulia. The 35 species described are as follows: G.acuminatum, N. Confervacea, M. granulata, P. Borealis, P. divergens, S. tenera, H. elongata, E. sudetica, F. construens, N. maharashtrensis, N. subtilis, N. paradox, F. zafarii, G. constrictum, C. gracilis, F. brevistriata, G. subtile, C. striata, N. productum, C. silicula, S. capronii, S. robusta, S. ulna, C. antique, M. juergensii, G. intricatum, S. capronioides, S. subsalsa, E. zebra, C. gracilis, S. tenera, F. jogensis, N. viriduloides, S. biseriata, N. kuetzingiana
CONCLUSION
The information obtained can be an aid in creating a reference database on diatoms for utilization in ecological assessment and identifying the location in the drowning cases.
ACKNOWLEDGEMENT
We would like to thank the Director, Institute of Forensic Science, Mumbai for the laboratory and library facilities. We acknowledge the immense help received from the scholars whose articles are cited and included in references of this manuscript. The authors are also grateful to authors / editors / publishers of all those articles, journals and books from where the literature for this article has been reviewed and discussed.
References:
1. Sarode P and Kamat N. Freshwater diatoms of Maharashtra. Aurangabad: Saikripa Prakashan. (1984)
2. Gandhi H. Some fresh-water diatoms from Lonavla Hill-Station in the Bombay-State (Maharashtra). Hydrobiologia 1962; 20(2):128-154.
3. Pareek R, Singh GP, Singh R. Some fresh water diatoms of Galta kund, Jaipur, India. Journal of Soil Science and Environmental Management 2011; 2(4):110-116.
4. Meeravali S, Prabhakara R, Venkata R. Floristic Studies on Diatom Flora from Selected Water Bodies of Ananthapuramu District Andhra Pradesh India. International Journal of Recent Scientific Research 2015; 6(10):6923-6930.
5. Rajvinder S, Rajinder S, Thakar MK. Extraction methods of diatoms-a review. Indian Internet Journal of Forensic Medicine and Toxicology 2006; 4(2).
6. Sasidharan A, Resmi S. Forensic diatomology-Review. Health Sciences 2014; 3(1). (accessed 16 March 2016).
7. Thakar MK, Rajvinder S. Diatomological mapping of water bodies for the diagnosis of drowning cases. Journal of Forensic and Legal Medicine 2010; 17. www.elsevier.com/jflm (accessed 30 November 2015).
8. Rajvinder S, Rajinder S, Thakar MK. Drowning associated Diatoms. Journal of Forensic and Legal Medicine 2005; 3(3). www. icfmt.net (accessed 30 November 2015).
9. Thakar MK, Guleria P. Tracking Drowning Trends in Himachal Pradesh during 2006-2010. Journal of Forensic and Legal Medicine 2015; 16(1). http://www.anilaggrawal.com/ij/vol_016_ no_001/papers/paper004.html (accessed 30 November 2015).
10. Singh R, Singh R, Kumar S, Thakar MK. Forensic Analysis of Diatoms- A Review. Journal of Forensic and Legal Medicine 2006; 7(2). http://www.anilaggrawal.com/ij/vol_016_ no_001/papers/paper004.html (accessed 2 August 2015).
11. Narayan P, Barupal G. A Systematic Account of the Fresh Water Diatom from Kaylana Lake of Jodhpur District, Rajasthan (India). International Journal of Recent Scientific Research 2015; 6(4).
12. Singh R, Deepa, Kaur R. Diatomological mapping of water bodies – A future perspective. Journal of Forensic and Legal Medicine 2013; 20(6).
13. Wikipedia, (2016). Diatom. [online] Available at: https:// en.wikipedia.org/wiki/Diatom
14. Www2.mcdaniel.edu, (2016). Diatoms. [online] Available at: http://www2.mcdaniel.edu/Biology/botsyl01/microalg/diatomsf/diatoms.html
15. Ucl.ac.uk, (2016). Diatoms. [online] Available at: http://www. ucl.ac.uk/GeolSci/micropal/diatom.html
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License