IJCRR - 4(6), March, 2012
Pages: 119-126
Print Article
Download XML Download PDF
AN INVITRO STUDY OF CISPLATIN INDUCED NEPHROTOXICITY IN GOAT KIDNEY
Author: N.Sangeetha
Category: General Sciences
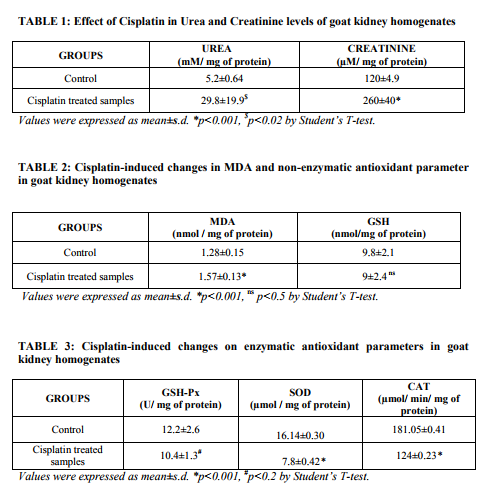
Abstract:Cisplatin (CP) is one of the most active cytotoxic agents in the treatment of cancer and has adverse side effects such as nephrotoxicity and hepatotoxicity. The present study was designed to determine the cisplatin induced nephrotoxicity by determining Urea, Creatinine, Malondialdehyde (MDA), reduced glutathione (GSH), Glutathione peroxidase (GPx), Superoxide dismutase (SOD) and Catalase (CAT) in goat kidney homogenates. A significantly (p< 0.02) increase in Urea and Creatinine was found. In addition, there was significant (p< 0.001) increase of MDA. But no significant difference in the non-enzymic antioxidant GSH in CP treated tissue homogenate but a less significant (p< 0.2) decrease in GPx and significant (p< 0.001) decrease in SOD and CAT was observed. Thus, excessive ROS production by CP causes an antioxidant imbalance which leads to lipid peroxidation and antioxidant depletion. In conclusion, CP treatment induces an irreversible kidney injury.
Keywords: Cisplatin, Nephrotoxicity, Oxidative stress, Antioxidant enzymes.
Full Text:
INTRODUCTION Cisplatin (cis-diamine-dichloroplatinum) is a prominent member of the effective broad spectrum antitumor drugs. However, its clinical usage is restricted due to some adverse side effects, such as ototoxicity and nephrotoxicity1-3 .However, at high doses, less common toxic effects, such as hepatotoxicity, may arise4-6 . Continued aggressive high-dose cisplatin chemotherapy necessitates the investigation of ways for prevention of the dose limiting side effects that inhibit the cisplatin administration at tumoricidal doses. In several studies, it had been documented that injection of CP produced a marked decrease in renal blood flow and glomerular filtration rate. The alterations in the kidney and liver functions induced by CP are closely associated with an increase in lipid peroxidation and reactive oxygen species (ROS) in the tissues 7, 8. Indeed, some recent studies have suggested that oxidative stress plays an important role in cisplatininduced liver damage 9-12 . ROS such as hydrogen peroxide, hydroxyl radical, singlet oxygen, superoxide anion and peroxyl radical are formed inside cells by exposure to several endogenous and exogenous agents, causing damage to many important biomolecules that have been implicated in several diseases13.These prooxidants are kept in check by endogenous antioxidants, but under disease conditions, the balance is shifted in favor of prooxidants, leading to oxidative stress. Excess ROS causes significant oxidative damage by attacking biomolecules such as membrane lipids, DNA and proteins in cells14 . The oxidative stress is associated with many disease states including neurological diseases such as Alzheimer‘s brains and Parkinson‘s disease, chronic heart disease, and kidney and liver diseases15 . Endogenous antioxidants such as reduced glutathione (GSH), glutathione peroxidase (GPx), superoxide dismutase (SOD), Catalase (CAT) are compounds that act as free radical scavengers. These antioxidants are electron donors and react with the free radicals to form harmless products such as water. Therefore, antioxidants protect against oxidative stress and prevent damage to cells16. Thus, the aim of the present study was to investigate the cisplatin-induced oxidative damages of goat kidney by biochemical methods. MATERIALS AND METHODS Sample Collection: The kidneys with intact capsules were taken from goat in the slaughter house approximately 15 to 20 min after electrocution; the renal artery and vein were flushed with ice-cold Eurocollins, pH 7.4, consisting of 177mM glucose, 10mM NaHCo3, 15 mM Kcl, 42 mM K2HPO4 and 15 mM KH2PO4 supplemented with 2 mM glycine. Freshly prepared homogenate were mixed with buffer (4mM Cacl2, 1.5% BSA). Experimental Design: The kidney homogenate samples were divided into 2 groups: Group I: Control (10% Kidney homogenate) Group II: 10% Kidney homogenate treated with 0.033 mM Cisplatin. Chemicals: Cisplatin was purchased from Dabur Pharmaceuticals Company. All the chemicals used were of analytical grade and were purchased either from Sigma chemical or Qualigens. Biochemical Measurements: Kidney homogenates were centrifuged for 15 min at 15000g and then clear supernatants were removed for analyses. The levels of Urea17 and Creatinine18 were determined and the results were expressed in mM/ mg of protein and µM/ mg of protein respectively. Malondialdehyde (MDA) level19 was measured and expressed as nmol / mg of protein. Measurement of tissue Glutathione (GSH)20, Glutathione peroxidase (GSHPx)21, Superoxide dismutase (SOD)22 , Catalase (CAT)23 activities were performed. The activities of antioxidant enzymes were expressed as nmol/ mg of protein for GSH, U/ mg of protein for GSH-Px, µmol / mg of protein for SOD and µmol/ min/ mg of protein for CAT. Statistical Analysis: Descriptive statistics were calculated for all the outcome variables and expressed as mean±s.d. The results were analyzed statistically according to the Student‘s ttest. The p values <0.02 were considered as significant. RESULTS Effect of Cisplatin in Urea and Creatinine levels: Table: 1 depicts the levels of urea and creatinine. Cisplatin treated samples showed significantly (p<0.02) increase levels to that of control samples. Cisplatin induced changes in MDA and GSH: Cisplatin treated kidney homogenate samples showed significant (p<0.001) increase in the levels of MDA and no significant difference in the non-enzymic antioxidant GSH comparing to control samples which has been portraited in Table: 2. Effect of Cisplatin on Enzymic antioxidant parameters: Studies on enzymic antioxidants GSH-Px showed less significant (p<0.2) decrease and SOD, CAT showed significant (p<0.001) decrease in Cisplatin treated samples compared to control as depicted in Table: 3. DISCUSSION Cisplatin, a platinum co-ordinated complex, is a widely used antineoplastic agent for the treatment of metastatic tumors of the testis, metastatic ovarian tumors, lung cancer, advanced bladder cancer and many other solid tumors24. The efficacy of Cisplatin is limited, however, by its doselimiting nephrotoxicity25-27. The treatment of tumor cells with CP provokes several responses including membrane peroxidation, dysfunction of mitochondria, inhibition of protein synthesis and DNA damage28, 29. Formation of free radicals leading to oxidative stress has been shown to be one of the pathogenic mechanisms of these side effects30 . The present work was undertaken to study detailed mechanism of CP-induced nephrotoxicity and it was confirmed that CP administration to the kidney homogenate of goat caused marked increase in urea and creatinine which is the diagnostic indicators of nephrotoxicity. Administration of CP (3mg/kg) to rats resulted in significant reduction of body weight and elevation of Urea and Creatinine levels31. Induction of nephrotoxicity by CP is assumed to be a rapid process involving reaction with proteins in the renal tubules32, 33. Because this renal damage occurs within 1 hr after administration34. It is important that the protective agent is present in renal tissue before damage occurs. The acute renal failure is indicated by increased urea and creatinine level before the development of tubular necrosis. These parameters are markers of glomerular filtration rate. However, it cannot be excluded that the enhancement of these parameters may be the result of tubular obstruction or tubular back leak35. It has been interpreted as a support of the hypothesis that tubular obstruction and tubular fluid back leak are not involved in the initiation of acute renal failure in this model of nephrotoxicity36 . Chronic treatment with cisplatin induced a highly significant increase in creatinine and significant increase in urea levels37 . Histologically, they observed the acute toxic tubular necrosis after CP administration which was similar to that produced in intoxication by mercury or cadmium38 suggesting that CP nephropathy may be attributable ultimately to the toxicity of the platinum molecules which induce damage in the proximal tubular cells. Lipid peroxidation was monitored by measuring MDA resulting from free radical damage to membrane components of the cells. A moderate increase in the MDA concentration was observed in the kidney tissue of rats treated with CP alone. Previous studies indicate an important role of ROS in the pathogenesis of the nephrotoxicity by CP 39-41. CP induced free radical production causing oxidative renal damage. However, in contrast to the former studies on acute nephrotoxicity induced by cisplatin in rats, little has been done to investigate the role of ROS in the development of chronic nephrotoxicity induced in rats by this drug42. But his study revealed no detectable renal production of ROS in rats with chronic renal failure, thus, the role of lipid peroxidation and its position in the chain of events that leads to Cisplatin nephrotoxicity still remains controversial. Various free radical scavengers have been shown to be effective in protection against CP- induced nephrotoxicity43 . The present study, CP showed significant increase in MDA. The mechanism by which Cisplatin causes renal damage is unclear, however, it has been postulated that oxidative stress is involved in this process 44-47 . Reduced glutathione has a multiple role as an antioxidant agent. It functions as a scavenger of ROS, including hydroxyl radicals, singlet oxygen, nitric oxide and peroxynitrite48. Data of our study indicate that GSH has no significant decrease after Cisplatin administration. Cisplatin may bind with the sulf-hydryl group of GSH and may substantially decrease the availability of GSH to scavenge free reactive oxygen metabolites. The Ciaplatin SH complex may also disrupt in lipid peroxidation and mitochondrial damage. Several studies demonstrated that cisplatin induced acute nephrotoxicity is mediated by depletion of renal GSH and by impaired activity of GPx as well as an increase in renal lipid peroxidation 49-53. Reactive Oxygen species (ROS) generated are immediately detoxified by major scavenger enzymes such as GPx, SOD and CAT. However, excessive production of ROS by Cisplatin causes antioxidant imbalance and leads to lipid peroxidation and antioxidant depletion. In our study, the major scavenger enzymes activities (GPx, SOD and CAT) were significantly decreased in kidneys of cisplatin treated samples. Our findings are similar to results of other investigators for kidney tissues which are incubated invitro with cisplatin caused low levels of antioxidant enzymes 54-56 . CONCLUSION In conclusion, our invitro study on cisplatin hypothesizes that, CP administration cause nephrotoxicity by suppressing antioxidant defense mechanism. Thus supplementation of vitamins during CP therapy may prevent damage to renal tubules by regulating oxidation- reduction potential of the cell. ACKNOWLEDGEMENT Authors acknowledge the immense help received from the scholars whose articles are cited and included in references of this manuscript. The authors are also grateful to authors / editors / publishers of all those articles, journals and books from where the literature for this article has been reviewed and discussed.
References:
1. Ek Born A, Lindberg A, Laurell G, Wallin I, Eksborg S, Ehrsson H, 2003. Ototoxicity, nephrotoxicity and pharmacokinetics of cisplatin and its monohydrated complex in the guinea pig. Cancer Chemotherapy and Pharmacology, 51: 36–42.
2. Iraz M, Kalcioglu MT, Kizilay A, Karatas E, 2005. Aminoguanidine prevents ototoxicity induced by cisplatin in rats. Annals of Clinical and Laboratory Science, 35(3): 329–35.
3. Yao X, Panichpisal K, Kurtzman N, Nugent K, 2007.Cisplatin nephrotoxicity: a review. The American Journal of Medical Sciences, 334(2):115–24.
4. Cvitkovic E, 1998. Cumulative toxicities from cisplatin therapy and current cytoprotective measures. Cancer Treatment Reviews, 24: 265– 281.
5. Hanigan MH, Devarajan P, 2003. Cisplatin nephrotoxicity: molecular mechanisms. Cancer Therapy, 1: 47– 61.
6. King PD, Perry MC, 2001. Hepatotoxicity of chemotherapy. The Oncologist, 6: 162–176.
7. Baligha R, Zhang Z, Baliga M, Ueda N, Shah SV, 1998. In vitro and in vivo evidence suggesting a role of iron in cisplatin-induced nephrotoxicity. Kidney International, 53: 394-401.
8. S. Silici, O. Ekmekcioglu, M. Kanbur, and K. Deniz, ?The protective effect of royal jelly against cisplatin-induced renal oxidative stress in rats,? World Journal of Urology, vol. 29, no. 1, pp. 127–132, 2010.
9. Lu Y, Cederbaum AI, 2006. Cisplatininduced hepatotoxicity is enhanced by elevated expression of cytochrome P450 2E1. Toxicological Sciences, 89: 515–523.
10. Iraz M, Ozerol E, Gulec M, Tasdemir S, Idiz N, Fadillioglu E, 2006. Protective effect of caffeic acid phenethyl ester (CAPE) administration on cisplatininduced oxidative damage to liver in rat. Cell Biochemistry and Function, 24(4): 357–61.
11. Pratibha R, Sameer R, Rataboli PV, Bhiwgade DA, Dhume CY, 2006. Enzymatic studies of cisplatin induced oxidative stress in hepatic tissue of rats. European Journal of Pharmacology, 532: 290-293.
12. Mansour HH, Hafez HF, Fahmy NM, 2006. Silymarin modulates cisplatin induced oxidative stress and hepatotoxicity in rats. Journal of Biochemistry and Molecular Biology, 39(6): 656–61.
3. B. Halliwell and J. M. C. Gutteridge, ?Lipid peroxidation: a radical chain reaction,? in Free Radicals in Biology and Medicine, B. Halliwell and J. M. C. Gutteridge, Eds., pp. 188–276, Oxford Clarendon Press, Oxford, UK, 1999.
14. K. A. Conklin, ?Dietary antioxidants during cancer chemotherapy: impact on chemotherapeutic effectiveness and development of side effects,? Nutrition and Cancer, vol. 37, no. 1, pp. 1–18, 2000.
15. J. T. Coyle and P. Puttfarcken, ?Oxidative stress, glutamate, and neurodegenerative disorders,?Science, vol. 262, no. 5134, pp. 689–695, 1993.
16. M. Valko, C. J. Rhodes, J. Moncol, M. Izakovic, and M. Mazur, ?Free radicals, metals and antioxidants in oxidative stress-induced cancer,? Chemico-Biological Interactions, vol. 160, no. 1, pp. 1–40, 2006.
17. Marsh WH, Fingerhut B, Miller H. Automated and Manual direct methods for the determination of blood urea. clin chem. 1965 jun;11:624–627
18. Bonsenes RW and Taussky HN (1945). On the colorimetric determination of creatinine by the Jaffe Reaction. J. Biol. Chem., 158: 587-591.
19. Hunter FE, Geblechi JM, Hofgten PE, Einstein S, Scott A. Determination of plasma malondialdehyde like materials. J Biol Chem 1963; 238: 614.
20. Moron MS, Depierre JW, Mannervk B. Levels of glutathione, glutathione reductase and glutathione-s-transferase activities in rat lung and liver. Biochem Biophys Acta 1979; 582: 67-68.
21. Rotruck JT, Pope AL, Ganther HE, Swanson AB. Selenium: Biochemical role as a component of glutathione peroxidase. Science 1973; 197: 588- 590.
22. Misra H and Fridovich I. The role of superoxide dismutase anion in the autooxidation of epinephrine and a simple assay for superoxide dismutase. J Biol Chem 1972; 247: 3170.
23. Aebi H. Catalase. In: Bergmeyer HV, editor. Methods in enzymatic analysis. Vol.2. New York: Academic Press, 1974:p.674-684.
24. Sweetman SC, 2002. Antineoplastic and immunosupressants. In: Sweetman, S.C. (Ed.), Martindale: The Complete Drug Reference, 33rd ed. Pharmaceutical Press, London, UK, pp: 525–527.
25. Winston JA, Safirstein R, 1985. Reduced renal flow in early cisplatininduced acute renal failure in the rat. The American Journal of Physiology, 249, F490– F496.
26. Greggi Antunes LM, Darin JD, Bianchi M, 2000. Protective effects of vitamin C against cisplatininduced nephrotoxicity and lipid peroxidation in adult rats. Pharmacological Research, 41: 405–411.
27. Chirino YI, Hernandez-Pando R, Pedraza-Chaveri J, 2004. Peroxynitrite decomposition catalyst ameliorates renal damage and protein nitration in cisplatin-induced nephrotoxicity in rats. BMC Pharmacology, 4: 20–29.
28. Cohen SM, Lippard SJ, 2001. Cisplatin: from DNA damage to cancer chemotherapy. Progress in Nucleic Acid Research and Molecular Biology, 67: 93-130.
29. Sadowitz PD, Hubbard BA, Dabrowiak JC, Goodisman J, Tacka KA, Aktas MK, Cunningham M.J, Dubowy RL, Souid AK, 2002. Kinetics of cisplatin binding to cellular DNA and modulations by thiolblocking agents and thiol drugs. Drug Metabolism and Disposition, 30: 183–190.
30. Jordan P, Carmo-Fonseca M, 2000. Molecular mechanisms involved in cisplatin cytotoxicity. Cellular and Molecular Life Sciences, 57: 1229– 1235.
31. Mahadev Rao, Mavinekere Marulapppa Kumar and Mysore Ananthanarayana Rao. In Vitro and In Vivo Effects of Phenolic Antioxidants against Cisplatin-Induced Nephrotoxicity, Journal of Biochemistry 1999; 125(2): 383-390.
32. Heidman HT, Gerkens JF, Jackson EK, Branch RA. Attenuation of cisplatin induced nephrotoxicity in the rat by high salt diet, furosemide and acetazolamide. Arch Pharmacol. 1985; 329:201–5.
33. Montine TJ, Borch RF. Role of endogenous sulfur containing nucleophiles in an in vitro model of cis-diamminedichloroplatiniuminduced nephrotoxicity. Biochem Pharmacol. 1990; 39:1751–7.
34. Rao M, Rao MM. Protective effects of selenomethionine against cisplatininduced renal toxicity in mice and rats. J Pharm Pharmacol. 1992; 50:687–91.
35. Jones TW, Chopra S, Kaufman JS, Flamenbaum W, Trump B. Cisdiammine-dichloroplatinium-induced acute renal failure in the rat. Lab Invest. 1985; 52:363–74.
36. Gholamreza Karimi, Mohammad Ramezani and Zahra Tahoonian. Cisplatin nephrotoxicity and protection by milk thistle extract in rats. Evid Based Complement Alternat Med. 2005;2(3):383-386.
37. Ricardo Ganzalez, Cheyla Romay, Aluet Borrego, Frank Hernandez, Nelson Merino, Zullyt Zamora and Enis Rojas. Lipid peroxides and antioxidant enzymes in cisplatininduced chronic nephrotoxicity in rats. Mediators Inflamm 2005 (3): 139-143.
38. Choie, D.D., Longnecker, D.S., delCampo, A.A., 1981. Acute and chronic cisplatin nephropathy in rats. Lab. Invest. 44, 397–402.
39. R. Baliga, Z. Zhang, M. Baliga, N. Ueda, and S. V. Shah, ?Role of cytochrome P-450 as a source of catalytic iron in cisplatin- induced nephrotoxicity,? Kidney International, vol. 54, no. 5, pp. 1562–1569, 1998.
40. Behling EB, Sendao MC, Francescato HDC, Antunes LMG, Costa RS and Bianchi MP (2006). Comparative study of multiple dosage of quercetin against cisplatin induced nephrotoxicity and oxidative stress in rat kidneys. Pharmacol. Rep., 58: 526-532.
41. Cetin R, devrim E, Kilicogula B, Avci A, Candir O and Durak I (2006). Cisplatin impairs antioxidant system and causes oxidation in rat kidney tissues: possible protective roles of natural antioxidant foods. J. Appl. Toxicol., 26(1): 42-46.
42. Trachtman H, Wilson D and Rao PS 1992 The role of oxygen free radicals in the development of chronic renal failure. Life Sciences., 50, 1877–1883.
43. Weichert-Jacobsen KJ, Bannowski A, Kuppers F, Loch T and Stöckle M (1999). Direct amifostine effect on renal tubule cells in rats. Cancer Res., 59: 3451-3453.
44. Inselmann G, Blomer A, Kottny W, Nellessen U, Hanel H, Heidemann HT: Modification of cisplatin-induced renal p-aminohippurate uptake alteration and lipid peroxidation by thiols, Ginkgo biloba extract, deferoxamine and torbafylline. Nephron 1995; 70:425- 429.
45. Yilmaz HR, Iraz M, Sogut S, Ozyurt H, Yildirim Z, Akyol O, Gergerlioglu S: The effects of erdosteine on the activities of some metabolic enzymes during cisplatin-induced nephrotoxicity in rats. Pharmacol Res 2004; 50:287- 290.
46. Mansour MA, Mostafa AM, Nagi MN, Khattab MM, Al-Shabanah OA. Protective effect of aminoguanidine against nephrotoxicity induced by cisplatin in normal rats. Comp Biochem Physiol Part C 2002; 132:123-128.
47. Baek SM, Kwon CH, Kim JH, Woo JS, Jung JS, Kim YK, Differential roles of hydrogen peroxide and hydroxyl radical in cisplatin-induced cell death in renal proximal tubular epithelial cells. J Lab Clin Med 2003; 142: 178- 186.
48. Halliwell B and Gutteridge JM (1989). Protection against oxidants in biological systems: the superoxide theory of oxygen toxicity. In: Free Radicals in Biology and Medicine (2nd ed.), Oxford, UK, Clarendon, pp.87- 187.
49. Somani SM, Husain K, Whitworth C, Rybak LP. Dose-dependent protection by lipoic acid against cisplatin-induced nephrotoxicity in rats: antioxidant defense system. Pharmacol Toxicol 2000; 86: 234_/240.
50. Saad SY, Najjar TA, Daba MH, AlRikabi AC. Inhibition of nitric oxide synthase aggravates cisplatin-induced nephro- toxicity: effect of 2-amino-4- methylpyridine. Chemotherapy 2002; 48:309–315.
51. Davis CA, Nick HS, Agarwal A: Manganese superoxide dismutase attenuates Cisplatin-induced renal injury: importance of superoxide. J Am Soc Nephrol 2001, 12:2683-2690.
52. Borrego A, Zamora ZB,Gonz´alez R, et al. Protection by ozone preconditioning is mediated by the antioxidant system in cisplatin-induced nephrotoxicity in rats. Mediators Inflamm. 2004; 13(1):13–19.
53. Gonzalez R, Borrego A, Zamora ZB, et al. Reversion by ozone treatment of acute nephrotoxicity induced by cisplatin in rats. Mediators Inflamm. 2004; 13(5-6):307–312.
54. A.A. Al-Majed, M. M. Sayed-Ahmed, A. A. Al-Yahya, A. M. Aleisa, S. S. Al-Rejaie, and O. A. Al-Shabanah, ?Propionyl-L-carnitine prevents the progression of cisplatin-induced cardiomyopathy in a carnitine-depleted rat model,? Pharmacological Research, vol. 53, no. 3, pp. 278–286, 2006.
55. M. Aleisa, A. A. Al-Majed, A. A. AlYahya et al., ?Reversal of cisplatininduced carnitine deficiency and energy starvation by propionyl-Lcarnitine in rat kidney tissues,? Clinical and Experimental Pharmacology and Physiology, vol. 34, no. 12, pp. 1252– 1259, 2007.
56. H. M. M. Arafa, ?Carnitine deficiency aggravates carboplatin nephropathy through deterioration of energy status, oxidant/anti-oxidant balance, and inflammatory endocoids,? Toxicology, vol. 254, no. 1-2, pp. 51–60, 2008.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License