IJCRR - 4(19), October, 2012
Pages: 31-36
Date of Publication: 15-Oct-2012
Print Article
Download XML Download PDF
FT-IR SPECTRAL ANALYSIS OF IMIDAZOLIUM CHLORIDE
Author: Seethalakshmi K., Jasmine Vasantha Rani E., Padmavathy R., Radha N.
Category: General Sciences
Abstract:IR spectra find wide spread applications for the qualitative and quantitative analysis of compounds and this technique is also well employed in the field of research to understand the nature of inter atomic bonding. In the present study, FT-IR spectra are recorded for 1-butyl-3-methyl imidazolium chloride (bmim)cl- and its solution in polar-protic solvent in the wave number region from 4000cm-1 to 450cm-1. The spectra are used to assign various stretching and bending modes of vibrations of the sample and also to identify the various phases present in the sample. A comparative study is made between the observed frequencies of the pure solvent and the solution. From this evaluation, the shifts in frequencies are corroborated. These shifts in the solution are ascribed to strong solute-solvent interaction by specific bonding between NH2 and C=O of solvent and Cl- and +NCH3 group of the imidazolium chloride.
Keywords: Ionic liquid, 1-Butyl-3-Methyl imidazolium Chloride, vibrational frequencies.
Full Text:
INTRODUCTION
Imidazolium chlorides are widely used as replacements for volatile organic solvents in industrial and laboratory processes as they are “environmentally benign”. They are non-volatile and non-flammable, with high thermal stability and show remarkable dissolution capabilities. Hence in the present investigation, solution of 1- butyl-3-methyl imidazolium chloride in formamide is analyzed for the specific bonding and interactions using IR spectral results.
RESEARCH METHODOLOGY
Experimental technique
1-butyl-3-methyl imidazolium chloride (bmim)cland formamide (99% purity AR Merck) were used as such.[1] The spectra are carried out in Sophisticated Analytical Instruments Facility (SAIF), IIT Madras. The IR spectroscopy is also carried out by using Fourier transform technique. The FTIR spectra of the compounds are recorded using PERKIN ELMER SPECTRUM ONE FTIR Spectrometer with a scan range MIR 4000 cm-1 - 450 cm-1 .This instrument has a typical resolution of 1.0 cm-1 .
RESULTS
The results of the current study are given under three headings.
Table.1 –Important stretching vibrations of Formamide
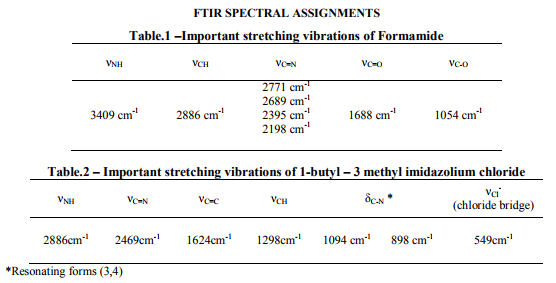
Formamide has two functional groups of importance that can be easily identified[2] by the FTIR Spectrum (figure1). The νNH stretching vibration, the νCH vibration, the νC=N vibration, the νC-O vibration are observed at various frequencies and it reveals that the formamide exists both in amide and imide forms.
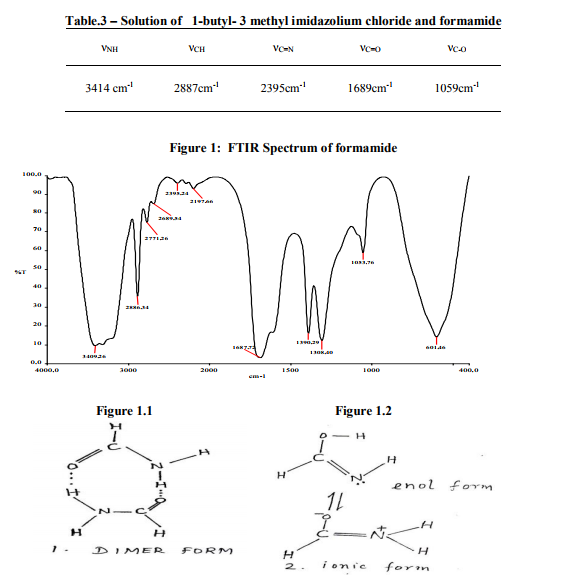
Table.2 – Important stretching vibrations of 1- butyl – 3 methyl imidazolium chloride
1-butyl-3-methyl imidazolium chloride exists in the solid state (figure 2). A series of peaks are observed and from the observation made, it is concluded that the change in vibrations are due to the resonating forms of the sample.
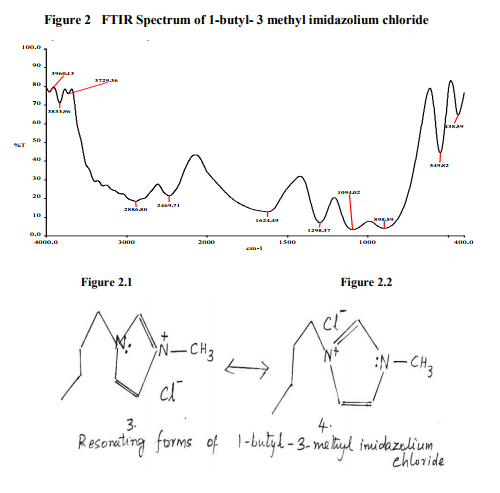
Table.3 –Important stretching vibrations of Solution of 1-butyl- 3 methyl imidazolium chloride and formamide
In the solution spectrum of 1-butyl-3-methyl imidazolium chloride and formamide, the important peaks are observed for various frequencies. The corresponding shifts in all the peaks obtained in the solution spectrum reveals the nature of the solute and the solvent.
DISCUSSION
FTIR Spectrum of formamide
At 3409 cm-1 νNH stretching vibration is observed (Table.1). At 2886 cm-1, a sharp peak of CH stretching vibration is found. At 2771 cm-1 and 2689 cm-1 , the stretching vibrations are assigned to the CH of the enolic form. A small amount of enolic form is identified by the νC=N at 2395 cm-1 and 2198 cm-1 vibrations.[3] The CO group appears as a sharp peak at 1688 cm-1 . The amide II bending modes are observed at 1390 cm-1 and 1308 cm-1 , νC-O vibration is observed at 1054 cm-1 . Thus, the amide exists as the dimer (figure 1.1) and the formamide exists both as the amide and imide forms (figure 1.2)[4] .
FTIR Spectrum of 1-butyl - 3 methyl imidazolium chloride
1-butyl-3-methyl imidazolium chloride in the solid state (figure 2) exhibits the characterization of a >N+CH3 group, the ammonium salt vibration at 2886 cm-1 . A series of peaks are observed in this region. (Table.2) At 2469 cm-1 , a broad absorption due to C=N is found. At 1624 cm-1 , C=C stretching vibration and CN bending vibration appeared as broad band. Further, at 1298 cm-1 , a sharp peak is observed due to CH bending vibration. At 1094 cm-1 and 898 cm-1 two bands of equal intensities are found[5,6]. This may be due to the resonating forms of the salt (figure 2.1 & 2.2). At 549 cm-1 , the CCl vibrations are observed.
FTIR Spectrum of Solution of 1-buty l- 3- methyl imidazolium chloride and formamide
The νNH vibration is observed at 3409 cm-1 in pure formamide and in the case of solution, the νNH vibration is changing from 3409 cm-1 to 3414 cm-1 (figure 3). A red shift of 5 cm-1 is observed in this solvent peak. (Table.3). The νCH vibration at 2887 cm-1 has not undergone much shift. Hence solvation has occurred at the N-H bond in formamide. The important peak corresponding to amide- I vibration is noticed at 1689 cm-1 (νC=O) has shifted only by 2 cm-1 . [7,8] The characteristic change in absorption frequency (νC-O) is found at 1059 cm-1 in the solution and in the pure solvent, it is found at 1054 cm-1 . The solution spectrum is thus indication of solvation at NH2 group (figure 3.1) and the enolate position of formamide (figure 3.2). This is further supported by the C=N vibration of formamide at 2395 cm-1 which is blue shifted (11 cm-1 ) to 2384 cm-1 in the solution.
CONCLUSION
- From the present study, FTIR spectral analysis leads to new insights about solvation.
- Strong solute-solvent interaction occurs in the non-aqueous solution of formamide and 1- butyl -3 methyl imidazolium chloride.
- Binding occurs between: NH2 and C=O of the solvent and Cland +NCH3 group of the imidazolium chloride through strong, coloumbic and ion-dipole interactions.
- A chloride ion bridge exists between solute and the solvent molecule.
ACKNOWLEDGEMENT
Author acknowledges the immense help received from the scholars whose articles are cited and included in references of this manuscript. The authors are also grateful to authors/editors/publishers of all those articles, journals and books from where the literature for this article has been reviewed and discussed. The author is highly thankful to the referees for their very constructive, valuable suggestions and useful technical comments, which led to a significant improvement of the paper.
References:
1. Wilkes, J.S.; Zaworotko, M.J. Air and water stable 1-ethyl-3-methylimidazolium based ionic liquids. Chem. Commun. 1992, 965-967.
2. Spectroscopic and thermodynamic analysis of some sodium salts in non- adequous medium. R.Padmavathy , E.Jasmine, K.Geetha, V.Solachi, N.Radha proceedings International Conference on Spectro Physics (INCONS 2005)
3. Mu, Z.G.; Wang, X.X.; Zhang, S.X.; Liang, Y.M.; Bao, M.; Liu,W.M. Investigation of tribological behavior of Al-Si alloy against steel lubricated with ionic liquids of 1- diethylphosphonyl-n-propyl-3- alkylimidazolium tetrafluoroborate. J. Tribol.- Trans. ASME 2008, 130, Art. No. 034501.
4. Qu,J.; Blau, P.J.; Sheng, D.; Huimin, L.; Meyer, J.M.; Truhan, J.J. Tribological characteristics of aluminium alloys sliding against steel lubricated by ammonium and imidazolium ionic liquids. Wear 2009, 267, 1226-1231.
5. Suzuki, A.; Shinka, Y.; Masuko, M., Tribological characteristics of imidazoliumbased room temperature ionic liquids under high vacuum. Tribol. Lett. 2007, 27, 307-313. Molecules 2009, 14,2906
6. Mazyar, O.A.; Jennings, G.K.; McCabe, C. Frictional dynamics of alkylsilane monolayers on SiO2: effect of 1-n-butyl-3- methylimidazolium nitrate as a lubricant. Langmuir 2009, 25, 5103-5110.
7. Minami, I.; Watanabe, N.; Nano, H.; Mori, S.; Kukumoto, K.; Ohno, H. Aspartic acid-derived wear preventing and friction reducing agents for ionic liquids. Chem. Lett. 2008, 37, 300- 301. Molecules 2009, 14 2907
8. Wasserscheid, P.; van Hal, R.; Bosmann, A. 1- n-Butyl-3-methylimidazolium ([bmim]) octylsulfate - an even 'greener' ionic liquid. Green Chem. 2002, 4, 400-404.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License