IJCRR - 9(6), March, 2017
Pages: 44-49
Date of Publication: 31-Mar-2017
Print Article
Download XML Download PDF
Evaluation of Microscopic Screening Methods for Detection of Urinary Tract Infection
Author: Akeela Fatima, Abiroo Jan, Nighat Akhter, Bashir A. Fomda, Mohd Suhail Lone, Junaid Ahmed, Lubna Samad, Shugufta Roohi
Category: Healthcare
Abstract:Objective: The study was conducted to evaluate the accuracy, cost and turnaround time of wet mount, gram stain and acridine orange stain microscopy of quantitative unspun urine for screening of urine samples. The combination of wet mount and gram stain and wet mount and acridine orange stain was also evaluated.
Materials and Methods: A total of 618 urine samples, which comprised of first ten consecutive samples that were received in the microbiology laboratory daily from patients with suspected urinary tract infection, were included in the study. All uncentrifuged urine specimens were subjected to wet mount, gram's stain, and acridine orange stain microscopy and semi quantitative urine culture tests. Time taken and expenditure for each test per sample was calculated and compared.
Result: Sensitivity, specificity, PPV and NPV of all the screening tests were compared singly and in combination. Acridine orange and gram stain had the highest sensitivity while wet mount, wet mount+gram stain and wet mount+acridine orange stain showed similar sensitivities. Specificity was highest for wet mount+acridine orange followed by acridine orange, wet mount+gram stain, gram stain and wet mount. PPV was highest for wet mount+acridine orange followed by acridine orange, gram stain, wet mount+gram stain and wet mount. NPV was highest for acridine orange followed by wet mount+acridine orange, gram stain, wet mount+gram stain and wet mount. All the microscopic tests were significantly rapid and cost effective as compared to culture test.
Conclusion: Acridine orange stain is recommended as a single screening test as it is an accurate, rapid and inexpensive method to rule out UTI.
Keywords: Wet mount, Urine microscopy, Acridine orange stain, Urine culture
Full Text:
INTRODUCTION
The term urinary tract infection encompasses a variety of clinical entities, including asymptomatic bacteriuria (ASB), cystitis, prostatitis, and pyelonephritis. Both symptomatic UTI and ASB connote the presence of bacteria in the urinary tract, usually accompanied by white blood cells and inflammatory cytokines in the urine. However, ASB occurs in the absence of symptoms attributable to the bacteria in the urinary tract and does not usually require treatment, while UTI has more typically been assumed to imply symptomatic disease that warrants antimicrobial therapy. Symptomatic UTI can be complicated or uncomplicated. Uncomplicated UTI refers to acute cystitis or pyelonephritis in nonpregnant outpatient women without anatomic abnormalities or instrumentation of the urinary tract and complicated UTI encompasses all other types of UTI.1 UTIs are the second most frequently occurring infections in the general population after upper respiratory tract infections.2 UTIs are the leading cause of gram negative sepsis in hospitalized patients and are the origin for about half of all nosocomial infections caused by urinary catheters.3 UTI is associated with considerable cost in terms of morbidity and economic and research expenditure.4
The most frequent cause of uncomplicated community acquired UTIs is Escherichia coli (E. coli) followed by Klebsiella spp., other Enterobacteriaceae, Staphylococcus saprophyticus, and enterococci. In complicated UTIs, the relative frequency of infection caused by Proteus, Pseudomonas, Klebsiella, and Enterobacter spp, increases. Hospitalized patients are most likely to be infected by E.coli, Klebsiella spp., Proteus spp., staphylococci, other Enterobacteriaceae, Pseudomonas aeruginosa, enterococci, and Candida spp.3 Since the last two to three decades UTIs due to multidrug resistant uropathogens have caused a growing concern worldwide. 5
Evaluation of suspected UTI includes history, physical examination and laboratory investigations. Urine culture and urine analysis for presence of pus cells and bacteria are important in the adequate management of UTI.6 Gold standard for diagnosis of UTI is quantitative urine culture for specific bacteria. However, this procedure is costly, and takes at least 24 hours; whereas an ideal test should be cheap, quick and should require only limited technical skill.7 Several rapid screening tests are used commonly to make a presumptive diagnosis of UTI, like dipstick biochemical analysis of urine for nitrites or leukocyte esterase (LE), catalase test, glucose oxidase test, automated assays and microscopic examination of urine for white blood cells by wet mount or for bacteria by Gram’s stain.8
Rapid diagnostic tests can rule out negative samples, are economical, save valuable time and thus are useful in high-end laboratories. Screening is also required in special circumstances where it is difficult to identify UTI on the basis of clinical criteria alone but where early diagnosis and prevention of complications affords significant benefit (e.g. children, and post renal transplant patients).9 As one diagnostic test is not reliable for confirmation of UTI, so researchers consider a combination of tests as the best choice for clinical decision making.7
The advantages to urine microscopy are that leukocytes, leukocyte casts, and other cellular elements are observed directly.10 Gram’s stain has added advantage of guiding the antibiotic therapy by observing the morphology and staining properties of the organisms. However, clinician should consider local sensitivity patterns of the possible pathogen.11 Acridine orange staining has also been evaluated as a microscopic bacteriuria screen as it is more sensitive and rapid method than Gram’s staining. 12
In the present study, we evaluated the accuracy, cost and turnaround time of wet mount, gram stain and acridine orange stain microscopy of quantitative unspun urine for screening of urine samples keeping semiquantitive urine culture as the gold standard for diagnosis of UTI. We further compared combination of wet mount and Gram’s stain and wet mount and acridine orange stain with culture for the same parameters.
MATERIALS AND METHODS
This prospective study was carried out in a tertiary care hospital from December 2015 to March 2016. The first ten consecutive urine samples that were received in the microbiology laboratory daily from patients with suspected urinary tract infection were included in the study. These comprised clean catch mid-stream, catheterized and suprapubic urine samples received in sterile containers.
A total of 618 samples were included in the study. All patient details were recorded along with urine sample collection methods. The samples were processed within one hour. All uncentrifuged urine specimens were subjected to wet mount, Gram’s stain, and acridine orange stain microscopy and semi quantitative urine culture tests.
- Direct microscopy or wet film preparation:
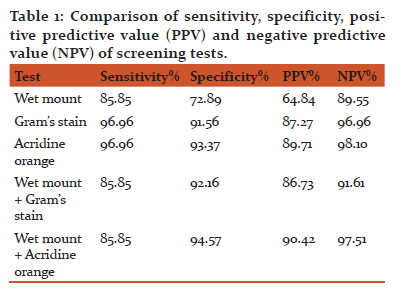
Fifty microlitre of well mixed uncentrifuged urine sample was placed on a clean, grease free, glass slide and covered with a 20 mm X 20 mm coverslip. The wet mount preparation was then examined under a high power magnification (40X) of a microscope for presence of pus cells. The presence of > 1 pus cell / 7 high power fields was considered significant pyuria.9
- Gram’s staining:
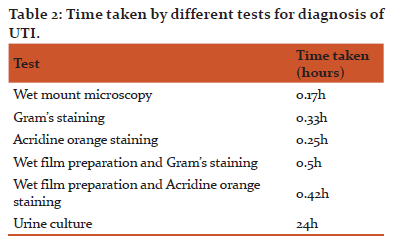
Fifty microlitre of well mixed uncentrifuged urine was smeared on a slide, air dried, heat fixed and Gram stained. At least 20 fields of the smear were scanned using oil immersion objective (100X). Presence of ≥1 bacterial cell per oil immersion field, which corresponds to 100,000 organisms/ml of urine, was considered significant.12, 13
- Acridine orange staining:
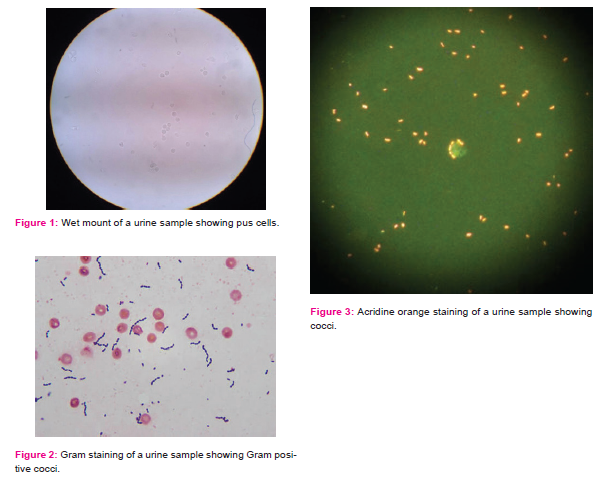
Hundred microlitre of well mixed uncentrifuged urine was smeared on a slide, air dried, fixed in 96% methanol for 2 minutes, flooded with acridine orange for 2 minutes, washed with distilled water, dried and examined under a fluorescent microscope. Presence of ≥ 1 morphologically similar orange coloured organism / 12 high power fields (40X) was considered significant. Presence of ≥ 2 morphologically different organisms indicated presence of mixed flora.12
- Urine culture and antimicrobial susceptibility testing:
Urine culture was routinely put up in the laboratory and identification of the organisms was done as per the standard procedure.
Turnaround time and cost analysis
Time taken by each test for one sample was calculated and compared. The cost of chemical constituents, consumables and other overhead charges was calculated for each of the screening tests and for culture and then compared.
Statistical Analysis
Diagnostic data were analyzed using 2 × 2 contingency tables. Sensitivity, specificity, positive predictive value (PPV), and negative predictive value (NPV) were calculated for each of the methods separately and in combination.
RESULTS
During the study period, a total of 2880 urine samples were submitted to the bacteriology laboratory for urine culture among which only 1400 showed significant microbial growth. As first ten consecutive urine samples daily were included in the study, only 618 samples from same number of patients were tested by microscopy.
Among the 618 urine samples analyzed, 266 (43.04%) were from male patients and 352 (56.95%) from female patients. Out of 618 samples, 382 (61.81%) were midstream clean-catch samples, 210 (33.98%) were catheter samples and 26 (4.07%) were suprapubic aspirates. The samples from OPD (out patient department) were 230 (37.21%) and from wards were 388 (62.78%). Out of 618 patients, 317 were having significant medical history, commonest being Type 2 Diabetes mellitus (16.82%) followed by sepsis (9.70%), hypertension (7.28%), recurrent UTI (4.85%), acute pancreatitis (3.72%), chronic kidney disease (2.58%), pregnancy (1.29%) and others (5.05%). Out of 342 patients, who were on antibiotics at the time of collection, 102 were culture positive.
Out of 618 samples, 332 (53.7%) were sterile, 198 (32%) showed significant microbial growth on culture, 22 (3.6%) yielded mixed growth and 66 (10.7%) showed growth of no significance. The results of 88 samples which showed mixed growth and growth of no significance were excluded from the study. Among the cultures with significant microbial growth, 68.68% (136/198) were from females and 31.31 % (62/198) were from males. Escherichia coli (48.48%) was the commonest isolate, followed by Candida spp. (18.18%), Enterococcus spp. (16.16%), Klebsiella pneumonia (9.09%), Acinetobacter baumanni (2.02%), Pseudomonas spp. (2.02%), Citrobacter spp. (1.01%), methicillin-resistant Staphylococcus aureus (1.01%) and combination of two organisms was present in 3.03%.
Among 198 culture positive urine samples, wet mount (Fig. 1) was positive in 170 samples, Gram’s stain (Fig. 2) was positive in 192 samples, acridine orange stain (Fig. 3) in 192 samples, wet mount + gram’s stain combination was positive in 188 samples and wet mount + acridine orange stain in 188 samples.
Sensitivity, specificity, PPV and NPV of all the screening tests were compared singly and in combination (Table 1). Acridine orange and Gram’s stain had the highest sensitivity while wet mount, wet mount+Gram’s stain and wet mount+acridine orange stain showed similar sensitivities. Specificity was highest for wet mount+acridine orange followed by acridine orange, wet mount+Gram’s stain, Gram’s stain and wet mount. PPV was highest for wet mount+acridine orange followed by acridine orange, Gram’s stain, wet mount+Gram’s stain and wet mount. NPV was highest for acridine orange followed by wet mount+acridine orange, Gram’s stain, wet mount+Gram’s stain and wet mount.
Comparison of turnaround time
Time taken by each test for processing of one sample was calculated and compared (Table 2). All the microscopic tests were significantly rapid as compared to culture test.
Cost analysis
The cost benefit was estimated as the difference between cost of culture and the rapid tests (Table 3). Cost of wet mount microscopy, Gram’s staining and acridine orange staining singly or in combination was more or less comparable to each other but when compared to urine culture was 15-40 times less.

DISCUSSION
In our study of 618 urine samples, 43.04% were from males and 56.95% from females. Among the urine cultures with significant microbial growth, 68.68% were from females and 31.31 % from males. This is in concordance to the increased prevalence of UTI in women; the main reason being anatomical and physiological differences between the two sexes.7
Out of 618 urine samples, 61.81% were midstream clean-catch samples, 33.98% were catheter samples and 4.07% were suprapubic aspirates. The ward: OPD ratio of submitted samples was 1.68: 1. Out of 618 patients, 317 were having significant medical history. The reason for such sample diversity and association with significant diseases was that all the patients were from a tertiary care hospital and were selected randomly.
In our study, Escherichia coli was the commonest organism isolated (48.48%), followed by Candida spp. (18.18%), Enterococcus spp. (16.16%), Klebsiella pneumonia (9.09%), Acinetobacter baumanni (2.02%), Pseudomonas spp. (2.02%), Citrobacter spp. (1.01%), methicillin-resistant Staphylococcus aureus (1.01%). Arslan S et al., in his study showed the growth of organisms on urine culture as; 47% Escherichia coli, 18.5% Klebsiella pneumonia, 10% Proteus mirabilis, and 8.5% staphylococci.14 Shobha KL et al., showed the isolation of organisms from culture in the sequence of E.coli 38%, Klebsiella spp. 8%, Enterobacter spp. 3%, Candida spp. 4%, Enterococcus spp. 22%, Citrobacter spp.1%, Acinetobacter spp.1%.15 Ali M et al., showed that organisms isolated from culture were in the order of; E.coli (65%) followed by Proteus spp.(16.3%), Pseudomonas spp.(7.6%), Enterococcus spp.(6.8%) and Klebsiella spp.(4.3%). 16 The higher prevalence of Candida in our isolates could be because more than 50% patients (342 out of 618) were on antibiotics.
As more than half of the urine samples routinely received in the laboratory during the study period (1400 out of 2880) showed no significant growth on culture, this results in unnecessary expenditure and delay in patient care. This can be overcome by screening tests which rule out negative samples, are economical, save valuable time and labor and thus are useful in high-end laboratories.
In the present study, three easily available and rapid tests were evaluated singly and in combination for their efficacy as screening tests. Wet mount examination of uncentrifuged urine was used to detect pyuria as it has been reported that wet mount of well mixed uncentrifuged urine is more reliable than that of centrifuged urine. 12 Significant pyuria, in the absence of significant bacteriuria in a symptomatic patient (e.g. acute urethral syndrome) is an indication for treatment and hence the importance of wet film examination.12 In this study, we evaluated this test taking ≥ 1 WBC / 7hpf 9,12 as cut off and found sensitivity of 85.85%, specificity of 72.89%, PPV of 64.88% and NPV of 89.55%. Taneja N et al, reported sensitivity of 68.4 %, specificity of 60.8 %, PPV of 32.7% and NPV of 87.3 % for this test.9 A study by Mohamed Ali et al. showed that this test has the sensitivity of 95.7%, specificity of 99.2%, PPV of 99.1% and NPV of 96.2%.16 A study by Cemal P et al showed that wet mount is 92% sensitive, 26% specific with 52% PPV and 78% NPV.2 Certain other previous studies have shown this test to have sensitivity ranging between 25-95% and specificity of 41-97 % but the PPV as low as 33 %.12 As most of the studies reported PPV of this test as low, this test, when used singly, cannot be relied upon as a screening test.
In the present study, Gram’s staining of urine samples showed sensitivity of 96.96%, specificity of 91.56%, PPV of 87.27% and NPV of 96.96 %. These values were in concordance with most of the previous studies. A study by Satish SP et al., reported sensitivity of 89.1%, specificity of 86%, PPV of 85.4% and NPV of 89.6% with this test.11 A study by Matias L et al., found that this test had sensitivity of 92.7%, specificity of 88.7, PPV of 68.5% and NPV of 97.9%.17 In a study by Amalia UP et al., this test had sensitivity of 88%, specificity of 100%, PPV of 100% and NPV of 90%.18 Thus, urine Gram’s stain is very reliable for screening of urine samples. In addition, Gram’s stain can guide the empirical treatment of patients with UTI.
Acridine orange stain as a urinary screening test has not been evaluated as much as Gram’s stain. Various studies have reported sensitivity from 92 to 98% and specificity from 59 to 87%.12 In the present study, we found that this test had sensitivity same as that of Gram’s stain i.e. 96.96% but a better PPV (89.71%), specificity (93.37%) and NPV (98.10%). Our results were in concordance with a study by Taneja N et al. who reported this test with a sensitivity of 90% and NPV of 98.8% but he reported lower specificity of 76.6%.12 Hoff et al. reported that this test had NPV of 99%.19 Based on the results of above studies and ours, it is inferred that when counts of >104 cfu/ml are taken as significant on culture, this test will eliminate the need for cultures in majority of the specimens. Thus, this technique can be recommended as a routine negative screening test especially in large laboratories.
In this study, combination of two tests, wet mount and Gram’s staining was also evaluated and we found sensitivity was 85.85%, specificity 92.16%, PPV 86.73% and NPV 91.61%. A study by Taneja N et al. showed that these two tests in combination had the sensitivity of 80%, specificity of 78.4%, PPV of 25% and NPV of 97.7%.12 A study by Matias L et al. showed that the combination of these two tests increased the sensitivity to 87.4%, specificity to 94 %, PPV 81.7%, and NPV 96.6%.17 A study by Sukru A et al., revealed that these two tests in combination had the sensitivity was 42%, specificity 90%, PPV 90% and NPV 40%.14 This combination had no advantage over gram stain as a screening test as sensitivity and PPV both decreased.
We also evaluated the combination of wet mount and acridine orange stain which to the best of our knowledge has not been done previously. When compared to culture, combination of two tests had sensitivity, specificity, PPV and NPV of 85.85%, 94.57%, 90.42% and 97.51% respectively. This combination had no advantage over acridine orange alone as a screening test. Thus, this study proved acridine orange stain to be the best screening test for diagnosis of UTI followed by Gram’s stain. Hence, absence of bacteriuria has reliable diagnostic value for ruling out UTI but positive results need to be confirmed by culture.
Negative predictive value is the probability that subjects with a negative test truly don’t have the disease. Our study indicated that acridine orange stain had NPV of 98%; that is strong for a screening test, and brings up property of this test to rule out UTI. This is important for a tertiary health care where a large number (more than 50% in our study) of cultures ordered routinely are negative. This could be attributed to prior use of antibiotics. In our patient population, 342 patients were on antibiotics at the time of collection of sample and out of these only 102 were culture positive.
Time taken by each test for processing of one sample was calculated and compared (Table 2), and all the microscopic tests were found to be significantly rapid than the culture test. Out of the microscopic tests, wet mount microscopy was the most rapid (0.17 h) followed by acridine orange stain (0.25 h) and Gram’s stain (0.33 h). As urine culture takes a minimum of 24 h to show any significant growth, microscopic tests serve as quite good screening tests for UTIs because of their rapidity; particularly in laboratories where a large percentage of urine cultures prove to be negative.
The cost benefit was estimated as the difference in expenditure on rapid tests and the culture (Table 3). The average cost of the screening tests was INR 1 as against INR 20 for the urine culture, amounting to a cost saving of 97.5%. Thus, the financial benefits of screening cannot be overemphasized. This cost factor makes these screening tests a viable and attractive option in peripheral centers where facilities of culture are not available and also in hospitals where majority of urine cultures prove to be negative.
Conclusion
Acridine orange stain is recommended as a single screening test as it is an accurate, rapid and inexpensive method to rule out UTI. As the need of a fluorescent microscope is a limitation to this test, Gram’s stain can be used as an alternative in laboratories where fluorescent microscope is not available.
Acknowledgements
We would like to thank the personnel of the bacteriology laboratory of the Sher-i-Kashmir institute of medical sciences, Soura, Srinagar, Kashmir. Authors acknowledge the immense help received from the scholars whose articles are cited and included in references of this manuscript. The authors are also grateful to authors / editors / publishers of all those articles, journals and books from where the literature for this article has been reviewed and discussed.
Ethical Clearance
The study has received ethical approval from Institutional ethical committee.
Source of funding
None.
Conflict of interest
None.
References:
- Kasper DL, Fauci AS, Hauser SL, Longo DLL, Jameson JL and Loscalzo J. Urinary Tract Infections, Pyelonephritis, and Prostatitis. In: Kalpana Gupta, Barbara W. Trautner. Harrisons principles of internal medicine. 19th edition. McGraw-Hill Education publications; 2005: 861.
- Plot C, Evliyaoglu O, Erkan REC, Ba?turk A, Tugrul Çak?r, Aslaner A et al. Comparison of urine microscopy and urine culture results of patients considered to have urinary tract infection. Am J Exp Clin Res. 2015; 2(3): 118-20.
- Betty AF, Sahm DF, Weissfeld AS. Bailey and Scott’s Diagnostic Microbiology, 11th edition. Philadelphia, Mosby publications; 2004: 927-38.
- Parajuli KP, Chaulagain DD, Singh G, Shrestha M, Chaudhary B, Surendra Chaurasia et al. Diagnosis of urinary tract infection: fresh urine microscopy or culture. Journal of Biomedical and Pharmaceutical Research. 2013; 2 (6): 63-64.
- Tiruneh M, Yifru S, Gizachew M, Molla K, Belyhun Y, Moges F et al. Changing Trends in Prevalence and Antibiotics Resistance of Uropathogens in Patients Attending the Gondar University Hospital, Northwest Ethiopia. International Journal of Bacteriology. 2014.
- Ninama AB and Shah PD. Comparison of various screening methods for presumptive diagnosis of significant bacteriuria. Int J Med Sci Public Health 2016; 5.
- Chenari MR, Gooran AS, Zarghami A and Faramarz Fazeli. Assessment of Urine Analysis Diagnostic Role: A Cross-Sectional Study in South Eastern of Iran Open Journal of Urology. 2012; (2): 227-231.
- Pezzlo M. Detection of Urinary Tract Infections by Rapid Methods. Clinical microbiology reviews. 1988; 268-280.
- Taneja N, Chatterjee SS, Singh M, Sivapriya S, Sharma M and Sharma SK. Validity of quantitative unspun urine microscopy, dipstick test, Leucocyte Esterase and nitrate tests in rapidly diagnosing Urinary Tract Infection. JAPI. 2010; 58.
- Hasan ASK, Kumar NT, Kishan RN and Neetha K. Laboratory diagnosis of urinary tract infections using diagnostics tests in adult patients. Int J Res Med Sci. 2014; 2(2): 415-42.
- Satish SP, Mahale K, Krishna S, Mariraj J, Anuradha SK and Sathyanarayan MS. Evaluation of gram stain of uncentrifuged urine as a screening method for diagnosis of urinary tract infections. Current Research in Medicine and Medical Sciences. 2011; 1(2): 19-21.
- Neelam T, Kavya M , Rungmei M, Nandita D and Meera S. Evaluation of three screening methods for detection of urinary tract infection in antenatal women. J Obstet Gynecol Ind. 2004; 54(3): 267-70.
- Shilpa K, Bernaitis L, Mathew J, Shobha KL, Ramyasree A and Manthena A
Comparison of urine microscopy and culture from urinary tract infection - a retrospective study. International Journal of Health Information and Medical Research. 2015; 2: 12-7.
- Arslan S, Caksen H, Rastgeldi L, Uner A, Oner AF and Odabas D. Use of Urinary Gram Stain for Detection of Urinary Tract Infection in Childhood. Yale journal of biology and medicine. 2002; 75: 73-8.
- Shobha KL, Rao AS, Shilpa K and Ramachandra L. Comparison of secreening test with urine culture in Urinary Tract Infection. International Journal of Medicine, 2014; 2(2): 81-3.
- Ali MM. Comparative Study of Dipstick, Urine Microscopy and Urine Culture in the Diagnosis of Urinary Tract Infection in Children Under Five Years. Karbala J. Med. 2010; 3(1): 822-28.
- Luco M, Lizama M, Reichhard C and Hirsch T. Urine microscopy as screen for urinary tract infection in a pediatric emergency unit in Chile: Pediatric Emergency Care 2006; 22 (10):705-09.
- Putri AU, Rina O, Rosmayanti, Ramayati R and Rusdidjas. Comparison of urine Gram stain and urine culture to diagnose urinary tract infection in children. Paediatr Indones. 2013; 53(2):121-4.
- Hoff RG, Newman DE and Staneck JL. 1985. Bacteriuria screening by use of acridine orange-stained smears. J. Clin. Microbiol. 21:513-16.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License