IJCRR - 5(5), March, 2013
Pages: 43-52
Date of Publication: 22-Mar-2013
Print Article
Download XML Download PDF
EFFET OF SODIUM CHLORIDE ON GROWTH OF HALOPHYTE BRUGUIERA GYMNORRHIZA (L.) Lamk.
Author: Manimegalai A., Manikandan T., Elumalai D.
Category: General Sciences
Abstract:Soil salinity is the major environmental stress and largely causes yield losses of crops worldwide. For the present study the halophyte seedlings of Bruguiera gymnorrhiza were selected. Seedlings were collected from mangrove forest in pichavaram, Tamilnadu, India. Seedling with uniform size were selected during September month and each seedling planted in 320 polytene bags separately .Before, each bags size with 12×18 cm were filled with sand, humus and red sand in 1:2:1 ratio. Various concentration of Sodium Chloride solution (0,100,200,300,400,500,600 and 700mM) were prepared by using distilled water. Plantation were separated into 8 groups, each group containing 40 number of plants. Each group of plants treated uniformly by different concentration of Sodium Chloride solution. In addition to this, plants were irrigated with distilled water and maintained as control (0). The results of investigation were made to study the effect of different concentrations of sodium chloride on growth of the seedlings of Bruguiera gymnorrhiza. The upper limits for the survival of the seedlings were up to 400 mM NaCl. The results indicated that the NaCl salinity stimulated the seedling growth upto the optimal level of salinity (i.e., 100mM and 400mM) and decreased significantly with increased salinity (500 to 700mM). The inhibition of seed growth was proportional to the concentrations of NaCl increased. The highest numbers of shoot length, root length were recorded at 400 mM NaCl concentration. Beyond 400 mM NaCl, the growth parameters increased drastically.
Keywords: Bruguiera gymnorrhiza, Conentrations, Growth, Halophyte, Increase, Decrease
Full Text:
INTRODUCTION
Soil salinity limits the plant growth and crop yield in many parts of the world, particularly in the arid and semi arid areas. Salinity tolerance of halophytes at germination varies among species (Yokoishi and Tanimoto, 1994; Liu et al. 2006). Salinity stress is a major limiting factor for plants germination and growth in coastal habitats as it is one of the most critical periods in life cycle of halophytes (Rubio Casal et al., 2003). Bruguiera gymnorhiza occurs along the coasts of tropical eastern and southern Africa, Madagascar and other Indian Ocean islands. Leaves decussately opposite, simple, and entire; stipules 4 cm long, reddish petiole 2–4.5 cm long. Bruguiera comprises 6 species, which are all used outside Africa in the same way as Bruguiera gymnorhiza. Medium sized, evergreen tree up to 36 m tall; bole up to 65 cm in diameter, buttressed; bark grey to almost black, roughly fissured, usually with large corky lenticels on buttresses and base of stem; knee roots or pneumatophores present. Saline soil is characterized by the presence of toxic levels of sodium and its chlorides and sulphates. Salinity tolerance is defined as the ability of plants to continuously grow under salt stress conditions (Munns, 2002). Halophytes, plants that survive to reproduce in environments where the salt concentration is around 200 mM NaCl or more, constitute about 1% of the world flora (Flowers and Colmer 2008).Halophytes as a group has one or more of several physiological adaptations that allow for the survival in the saline environment. Halophytes will not only offer great potential as novel crops but also important models for understanding salt tolerance in plants. A halophyte is a plant which is capable of surviving in a highly salty environment. Halophytes actually have increased growth at low salt concentrations (compared to no salt), with decreased growth at much higher concentrations. Plant growth and productivity is severely affected by high salinity. According to Maggio et al., 2010, high levels of soil salinity can cause water deficit, ion toxicity, and nutrient deficiency leading to molecular damage and even plant death. Plants have evolved a variety of protective mechanisms to allow with these unfavorable environmental conditions for survival and growth including the accumulation of ions and osmolytes such as proline.Some grasses could grow in the soil salinity ranges between 300 to 500 mM NaCl ( Peng et al., 2004) while others could not survive in the salt concentration above 300 mM NaCl (La Peyre and Row, 2003).Growth of some halophytes is generally stimulated by various levels of salinity(Pujol et al., 2001).Salt stress severely limits the plant growth and yield; in fact no toxic substance restricts the plant growth more than salt on world scale (Xiong and Zhu, 2002). Growth of S. ioclados is inhibited with an increase in salinity. Most halophytic grasses do not survive in more than 300 mM NaCl (Glenn, 1987). Optimal growth of some monocotyledonous halophytes was observed in 300 mM NaCl (Breen et al., 1977;Lissner and Schierup, 1997).Bruguiera gymnorrhiza is a common mangrove tree which supports as a wall of mangrove forest and is widely distributed in Thailand and southeast Asia, southern and eastern Africa, Australia, Micronesia and Polynesia (Hou, 1970). Halophytes actually have increased growth at low salt concentrations (compared to no salt), with decreased growth at much higher concentrations. Plant growth and productivity is severely affected by high salinity. In the present investigation an attempt has been made to study the effect of Sodium Chloride on germination constituents of Bruguiera gymnorrhiza. Typically, it is most common in the middle and upper intertidal zones, rather than in the lower intertidal zone or along the seaward edge of mangrove stands. It is a medium to tall tree that may reach 30–35 m (100–115 ft) in height, although it is commonly much shorter. Diameters are commonly about 15–35 cm (6–14 in). Leaf color, size, and shape enable B. gymnorrhiza trees to be distinguished from other Bruguiera spp. As such, planting simply entails gently pushing the distal end of the 10–15 cm (4–6 in) long hypocotyl one third of its length into the sediment, spaced at about 1 m (3.3 ft) intervals. Leaves and roots may begin to develop within a week or two of sowing. Propagation is simple and relies on the special feature of this genus: viviparous propagules
MATERIALS AND METHODS
Plant material: Halophytic plant Bruguiera gymnorrhiza was selected for the present study, commonly large-leafed mangrove called as Oriental or Black mangrove belongs under Rhizophoraceae family. Collection of Plant Material Study Area: For the present investigation Bruguiera gymnorrhiza has been collected from the mangrove forest in Pichavaram in Tamilnadu. In India there are 7,00,000 ha of area covered by mangroves along the estuaries and major deltas. Indian mangroves comprised of more than 60 species belonging to 41 genera and 29 different families. Pichavaram mangrove forest is located in the southeast coast of India, at about 225 km south of Chennai and 5 km north east of Chidambaram, Cuddalore district, Tamil Nadu, between latitude 11°20' to 11°30' north and longitudes 79°45' to 79°55' east. Mangrove is present in the higher land of Vellar - Coleroon estuarine complex. The mangrove extends to an area of 1,100 hectares, representing a heterogeneous mixture of mangrove elements. The mangrove comprises about 51 small and large islands with their sizes ranging from 10 m²to 2 km².Two major rivers viz. Vellar and Coleroon drain into the Bay of Bengal in this area. The area between the two rivers has brackishwater with mangrove vegetation. During 90s, M.S. Swaminathan Research Foundation (MSSRF), Chennai, India established a mangrove Genetic Resource Conservation Centre here by adopting 50 ha forest area. Plant establishment: Equal-sized Seedlings were collected from coastal region of mangrove forest in pichavaram, during the month of September, 2010. The polythene bags size with 12×18 cm filled with homogenous mixture of garden soil containing red earth, sand and farmyard manure (1:2:1). Seedling with uniform size were selected during September month and each seedling planted in 320 polythene bags separately. Plantation were separated into 8 groups and each group containing 40 number of plants. Various concentration of Sodium Chloride (0,100,200,300,400,500,600,700mM) Salt solutions were prepared with NaCl (Laboratory Grade, Glaxo Laboratories, India), by using distilled water and it used for the present study. Each group of plants treated uniformly by different concentration of Sodium Chloride solution. In addition to this, plants were irrigated with Distilled water and maintained as control (0).All the experiments were conducted in the PG and Research, Department of Plant Biology and Biotehnology, Arignar Anna Government Arts College, Villupuram, Tamilnadu. The treatments were continued until the plants received the required concentrations of the salt, after this all the plants were irrigated with tap water. The experimental yard was roofed with transparent polythene sheet at the height of 3 m from the ground in order to protect the plants from rain. Sampling for various studies was taken on the 30th day after NaCl treatment. Plantations of the study area were continuously treated with various concentrations of NaCl solution and growths of plants were recorded in 30, 60, 90 and 120 days after sowing regularly. Growth Study: Shoot length (cm plant-1). Plant height was recorded by measuring the height of the plant from the surface of the soil to the tip of the top most leaf. This was recorded on 30,60,90 and120 days after treatment and expressed in cm plant-1. Root length (cm plant-1) The root length was measured from the point of first cotyledonary node to the tip of longest root and expressed in cm plant-1.
RESULTS AND DISCUSSION
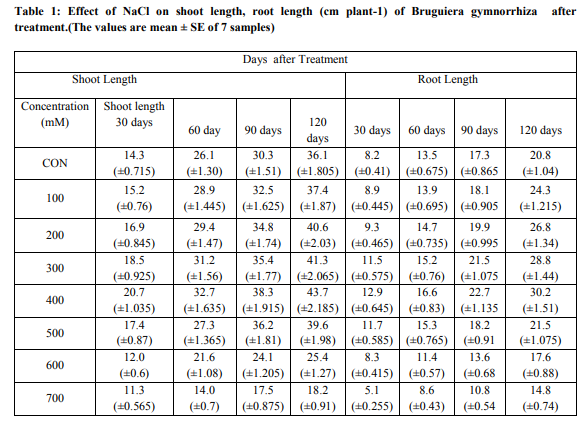
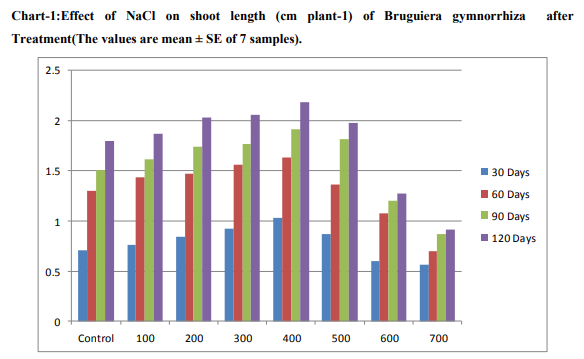
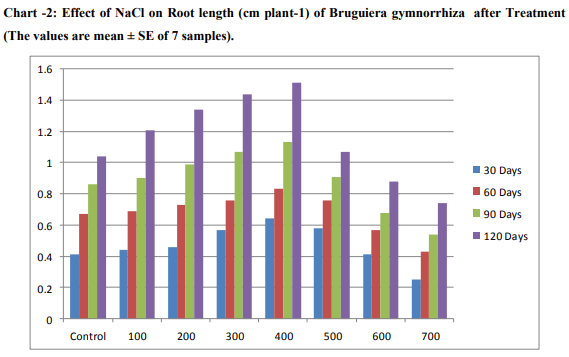
Effect of salinity on growth The effect of different concentrations of NaCl on the shoot length per plant is presented in Chart (1 and 2). The maximum increase in shoot length was observed on the 400mM NaCl. In the optimum concentration, the calculated shoot length was high when compared to that of control. Beyond this optimum concentration, there was a gradual reduction in length of shoot was noticed.Plant height decreased with increasing salinity for halophyte Bruguiera gymnorrhiza. Plant Shoot and root length were decreased in the plants supplemented with NaCl and their rate of loss was proportional to the concentration of the NaCl. Saline soils and saline irrigations constitute a serious production problem for vegetable crops as saline conditions are known to suppress plant growth (Shannon, et al., 1999). The results from Chart -1 .on growth of B.gymnorrhiza shows that higher shoot length (± 1.035) in 400mM treatment (±0.715) than control in 700mM treated seedling on 30th day and 400mM (±1.635),control (±1.305) on 60th observations .Other both 90 and 120 days observations increased seedling growth (±1.915),( ±2.185) in 400mM treatment respectively. Present findings supports the other researches.Greipsson and Davy (1996) also reported differences in salt tolerance of Leymus arenarius seedlings grown with seeds from inland and coastal populations. Seedlings of coastal origin had higher number of tillers at 200-400mM NaCl, while dry matter production was less adversely affected at higher salinities. In general, low salinity levels do not appear to have a deleterious effect on the growth of Atriplex spp. and may actually stimulate growth (Matoh,et al.,1986).According to Curtis,et al.,1986, the reduction of shoot growth and leaf area development of A.hortensis with increasing salt concentration in Kenaf (Hibiscus cannabinus) under moderate salt stress was affected primarily through a reduction in elongation of stem and leaf area development. Similarly,the results from Chart -2, increased root growth of seedlings in 400mM (±0.645) 0n 30 days treatment .It gradually increased and the increased values (±1.51) obtained in 120th day than control.The increased values gradually decreased in 500mM upto 700mM treatment both in shoot and root length.Low shoot length values were (±0.565)obtained in 30days,( ±0.7) in 60 days,( ±0.875) in 90 days and Very low growth value (±0.91)in 120 days treated seedlings.At high salinities, growth reduction might be caused by a reduced ability to adjust osmotically, as a result of saturation of solute uptake system or excessive demand on the energy requirement of such systems (Gale,et al.,1985).Growth of S. ioclados is inhibited with an increase in salinity. Most halophytic grasses do not survive in more than 300 mM NaCl (Glenn, 1987). Optimal growth of some monocotyledonous halophytes was observed in 300 mM NaCl (Breen et al., 1977)Very declined growth values of root length (±0.255)in 30th day and (±0.74) in 120th day treated seedlings of Bruguiera gymnorrhiza. Previous studies in Atriplex amnicola also indicated that this plant increased growth after additions of NaCl to the growth medium up to 25-50 mM, but then growth declined as salt concentration was increased(Aslam,et al.,1986).
DISCUSSION
Halophytes shape the environments they live in, changing the surface of the Earth in the process. Certain halophyte species can also be used as animal fodder, an advantage in some communities. The results from Chart 1 and 2 on the various concentrations of NaCl solution treatment on seedling germination of Bruguiera gymnorrhiza observations revealed both positive and negative effects are obtained on these Bruguiera gymnorrhiza halophytic plant. The shoot and root length of seedlings of Bruguiera gymnorrhiza slowly increasing from 100mM upto 400mM concentrations of NaCl solution. From the results seedling growth of Bruguiera gyhmnorrhiza was maintained up to 400 mM NaCl. According to Manimegalai et.al.,(2012) seedling Germination activity of Bruguiera gyhmnorrhiza was maintained up to 400 mM NaCl. NaCl had increased the number of seedling germination with increasing concentrations up to 400mM. However, the presence of high salt levels does not seem to be required for optimal growth. It is reported that soil salinity suppresses shoot growth more than the root growth (Ramoliya,et al.,2004). The present findings strongly supports by some of the researches. Most of the plants cannot tolerate high salt concentrations of the soil and cannot be grown on a salt affected land (Glenn and Brown, 1999). Kelly et al.(1982), Daoud et al.(2001) and Harrouni et al. (2001) who reported that low NaCl concentrations stimulate growth of some halophytic species, but an excess of salt decreases growth and biomass production. The growth of Sesuvium portulacastrum showed positive effect to NaCl concentrations upto 600 mM and the upper limit for survival of this species was 900 mM (Ashraf, M., 1999.).The optimum growth for seedlings observed in 400mM,while dereasing the shoot and root length above this optimum level of increasing concentrations 500,600 and 700mM of NaCl solution.Similar findings supports present studies that reports the germination percentage is reduced with a high NaCl concentrations (Tobe et al., 2001; Rubio-Casal et al., 2003).Salt stress affects germination percentage, germination rate and seedling growth in different ways depending on the plant species (Ungar, 1996; Gul and Weber, 1999 ). Salinity has been shown to be one of the external factors that influence the process of senescence and the consequent shedding of leaves (Pool et al., 1975). At higher concentrations, the fresh weight of leaf, stem and root was reduced. The increase in fresh weight of the leaf tissues can be attributed to the increase in leaf thickness and the accumulation of ions and water in the tissues (Khan et al., 2005). Physiological disorders such as reduced growth are ultimately due to the cumulative effects of the causal factors on the physiological processes necessary for plant growth and its development (Schutzki and Cregg, 2007). Exposure to high NaCl imposes oxidative stress due to changes in the osmotic and ionic environment in plant (Alakhverdiev et al.2000).Our results indicated that sodium and chloride concentration in shoots and roots increased with salinity. It would appear that the growth response at moderate salinities may be largely the consequence of an increased throughput of solutes required to derive cell expansion—although this does not result in increased turgor pressure.Growth analysis Mean relative growth rate was stimulated by moderate external salinity, reaching a peak at 400mM NaCl that was nearly double the value in the absence of NaCl (Chart-1 and 2). Further increase in salinity caused a reduction in growth to a very low value at 700 mM NaCl. Total growth after 60 d of salinity treatment was also greatest in the 400 mM NaCl treatment and was highly correlated with decreasing concentration. Among the different concentrations of NaCl treatment, the 400mM NaCl alone showed a promontory effect while the other concentrations exhibited inhibitory effect on the germination of Bruguiera gymnorrhiza seedlings. The intensity of inhibition was proportional to the concentration of the NaCl concentration increased.
CONCLUSION
Results from investigation shows the inhibition of seedling shoot and root length in Bruguiera gymnorrhiza was caused by the NaCl solution . The plants that can be naturally established in saline soils called halophytes. When increasing the concentration from 100 upto 300mM the seedling growth was influenced. The concentration of 400 mM NaCl solution alone highly promoted the seedling growth .While increasing the other concentrations 500,600,700mM inhibited and delayed seedling germination of Bruguiera gymnorrhiza over control intensity of inhibition. Saline soil is characterized by the presence of toxic levels of sodium and its chlorides and sulphates. Increasing the concentration of NaCl with increasing the rate of shoot and root length of seedlings ,while decreasing the concentrations of NaCl with decreasing the rate of seedling shoot ,root length of Bruguiera gymnorrhiza. Maximum growth rate shows in 400mM and very low and negative growth rate obtained in 700mM of NaCl. Maas (1987) reported that in most halophytic species growth decreases gradually with the increase of salt rate in the culture medium above a critical threshold specific to each species. It was reported that maximum germination of the seeds of halophytic plants occurred in distilled water or under reduced salinity ( Carter and Ungar, 2003).The intensity of inhibition of growth was proportional to increasing the concentrations of NaCl solution employed.Positive growth proportional to low concentration and negative growth proportional to higher concentrations.
References:
1. Allakhverdiev S.I., Sakamoto A., Nishiyama Y., Inaba M., Murata N. (2000): Ionic and osmotic effects of NaClinduced inactivation of photosystems I and II in Synechococcus sp. Plant Physiol., 123: 1047–1056.
2. Aslam, Z., Jeschke, W.D., Barrett-Lennard, E.G., Setter, T.L., Watkin, E., Greenway, H. (1986). Effects of external NaCl on the growth of Atriplex amnicola and the ion relations and carbohydrate status of leaves. – Plant. Cell. Environ. 9: 571-580.
3. Ashraf, M. (1999). Breeding for salinity tolerance in plants. Crit. Rev. Plant Sci., 13: 17-42.
4. Breen, C.M., C. Everson and K. Rogers. (1977). Ecological studies on Sporobolus virginicus (L.) Kunth with particular reference to salinity and inundation. Hydrobiologia, 54: 135–140.
5. Curtis, P.S., Lauchli, A. (1986): The role of leaf area development and photosynthetic capacity in determining growth of kenaf under moderate salt stress. – Austral. J. Plant Physiol 13: 553-565.
6. Carter, C.T. and I.A. Ungar.(2003). Germination response of dimorphic seeds of two halophyte species to environmentally controlled and natural conditions. Can. J. Bot., 81, 918-926.
7. Daoud S., Koyro H-W. and Harrouni, M.C. (2001). Salinity tolerance of beta vulgaris ssp. Maritima. I. Biomass production and osmotic adjustment. (to appear in H. Lieth, Ed. "Cash Crop halophytes : Potentials, pilot Projects, Basic and Applied Research on Halophytes and saline irrigation. Ten years after the Al Ain meeting" T:VS Series, Kluwer academic publishers group, the Netherlands).
8. Flowers TJ, Colmer TD (2008) Salinity tolerance in halophytes. New Phytol 179:945–963
9. Glenn, E.P.(1987). Relationship between cation accumulation and water content of salt-tolerant grasses and a sedge. Plant, Cell Environ., 10: 205–212.
10. Greipsson, S. and A. Davy. (1996). Sand accretion and salinity as constraints on the establishment of Leymus arenarius for land reclamation in Iceland. Ann. Bot., 78: 611– 618.
11. Gale, J.; Zeroni, M.(1985) Response of Plants to Salinity I Desert Controlledenvironment Agriculture (CEA): A Model for Plant–Salinity–Environment Interactions. In Proceedings of Third Biosalinity Workshop; Pasternak, D., San Pietro, A. Eds.; W. Junk: The Hague, 57– 68.
12. Glenn, E.P. (1987). Relationship between cation accumulation and water content of salt-tolerant grasses and a sedge. Plant, Cell Environ., 10: 205–212.
13. Glenn EP, Brown JJ .(1999). Salt tolerance and crop potential of halophytes. Crit. Rev. Plant Sci., 18: 227-255.
14. Gul, B. and D.J. Weber.(1999). Effect of salinity, light, and temperature on germination in Allenrolfea occidentalis. Can. J. Bot., 77, 240-246.
15. Hou D (1970). Rhizophoraceae. In: Flora of the Thailand Smitinand T. Larsen K. editors. Bangkok. Asret Press, pp. 5-15.
16. Harrouni M.C., Daoud S. and Koyro HW.(2001). Effect of seawater irrigation on biomass production and ion composition of 7 halophytic species in Morocco. (to appear in: H. Lieth, Ed. "Cash Crop halophytes : Potentials, pilot Projects, Basic and Applied Research on Halophytes and saline irrigation. Ten years after the Al Ain meeting" T:VS Series, Kluwer academic publishers group, the Netherlands).
17. Kelly,D.B., J.R. Goodin, andD.R. Miller,(1982). Biology of Atriplex, Chap. (5) Tasks for vegetation. Sc., 2. D.N. Sen and K.S. Rajpurohit (eds.).
18. Khan, M.A., I.A. Ungar and A.M. Showalter,(2005). Salt stimulation and tolerance in an inter tidal stem-succulent halophyte. J. Plant Nutr., 28: 1365-1374.
19. Lissner, J. and H.H. Schierup.(1997). Effects of salinity on the growth of Phragmitis australis. Aquat. Bot., 55: 247– 260.
20. Le Peyre, M.K. and S. Row.(2003). Effects of salinity changes on growth of Ruppia maritima L. Aquat. Bot., 77: 235-241.
21. Liu, X., H. Qiao, W. Li, T. Tadano and M.A. Khan.( 2006).Comparative effect of NaCl and Seawater on seed germination of Suaeda salsa and Atriplex centralasiatica. In: Biosaline agriculture and salinity tolerance in plants. M. Ozturk, Y. Waisel, M.A. Khan and G. Gork (eds.). Birkhauser Verlag, Switzerland, pp: 45-53.
22. Maas, E.V., Grieve, C.M.(1987). Sodium induced calcium deficiency in salt-stressed corn. – Plant Cell and Environment 10: 559-564.
23. Maggio A, Barbieri G, Raimondi G, De Pascale S (2010). Contrasting effects of GA3 treatments on tomato plants exposed to increasing salinity. J Plant Growth Regul 29:63–72.
24. Munns, R.(2002).Comparative physiology of salt and water stress. Plant Cell Environ., 25: 239-50.
25. Matoh, T., Watanabe, J., Takahashi, E. (1986).Effects of sodium and potassium salts on the growth of a halophyte Atriplex gmelini. – Soil Science and Plant Nutrition 32: 451-459.
26. Peng, Y.H., Y.F. Zhu, Y.Q. Mao, S.M. Wang, W.A. Su and Z.C. Tang.(2004). Alkali grass resists salt stress through high [K+] and endodermis barrier to Na+. J. Exp. Bot., 55: 939–949.
27. Pool, D.J., A.E. Lugo and S.C. Snedaker,(1975). Litter production in mangrove forest of Southern Florida and Puerto Rico. In: Proc. Int. Symp. Biol. and Management of mangroves. Vol. I. Walsh, G.E. S.C. Snedaker and H.J. Teas. (eds.), pp. 213-237. Int. Food. Agri. Sci, Univ. Florida, Gainesville.
28. Pujol, J.A., J.F. Calvo and L. RamirezDiaz.(2001). Seed germination, growth and osmotic adjustment in response to NaCl in a rare succulent halophyte from southeastern Spain. Wetlands, 21: 256-267.
29. Ramoliya, P.J., Patel, H.M., Pandey, A.N. (2004). Effect of salinization of soil on growth and macro and micro nutrient accumulation in seedlings of Acacia catechu (Mimosaceae). – Annals of Applied Biology 144: 321-332.
30. Rubio-Casal, A.E., J.M. Castillo, C.J. Luque and M.E. Fig.ueroa.(2003).Influence of salinity on germination and seeds viability of two primary colonizers of Mediterranean salt pans. J. Arid Environ., 53, 145-154 .
31. Schutzki, R.E. and B. Cregg.( 2007). Abiotic Plant Disorders Symptoms, Signs and Solutions. A Diagnostic Guide to Problem Solving. Extension Bulletin. E2996.
32. Shannon, M.C., Grieve, C.M. (1999): Tolerance of vegetable crops to salinity. – Sci. Hortic. 78(1/4): 5-38.
33. Tabaee Oghdaee, R.(1999). Environmental tolerance potency in some rangeland grasses. Journal of Pajouhesh and Sazandegi, 40: 41-45.
34. Tobe, K., L. Zhang, G.Y. Qui, H. Shimizu and K. Omasa.(2001) Characteristics of seed germination in five non-halophytic Chinese desert shrub species. J. Arid Environ., 47, 191-201.
35. Yokoishi, T. and S. Tanimoto,(1994). Seed germination of the halophyte Suaeda japonica under salt stress. J. Plant Res., 107: 385-388.
36. Ungar, I.A.(1996). Effects of salinity on seed germination, growth and ion accumulation of Atriplex patula (Chenopodiaceae). Am. J. Bot., 83, 604- 607.
37. Xiong L, Zhu JK (2002). Molecular and genetic aspects of plant responses to osmotic stress. Plant Cell Environ., 25: 131- 139.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License