IJCRR - 8(23), December, 2016
Pages: 11-15
Print Article
Download XML Download PDF
A STUDY OF THYROID PROFILE AND SERUM ALBUMIN IN PREECLAMPSIA WOMEN
Author: Rahul R. Chaudhary, M.G. Muddeshwar
Category: Healthcare
Abstract:Aim: The study was carried out to estimate thyroid profile and serum albumin level in preeclamptic women as compare to normotensive women. In addition to this correlation between thyroid profile and albumin in preeclamptic women was also calculated.
Material and Methods: The study comprised of 60 cases of preeclamptic women who attended in Obstetrics and Gynaecology Department in Govt. Medical College, Nagpur from February 2013 to August 2014 and 60 normotensive healthy womens as controls. Serum total Tri-iodothyronine (T3), Tetraiodothyronine (T4), Thyroid Stimulating Hormone (TSH) and serum albumin were estimated.
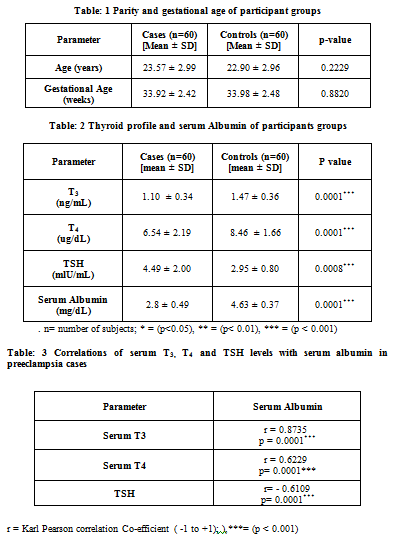
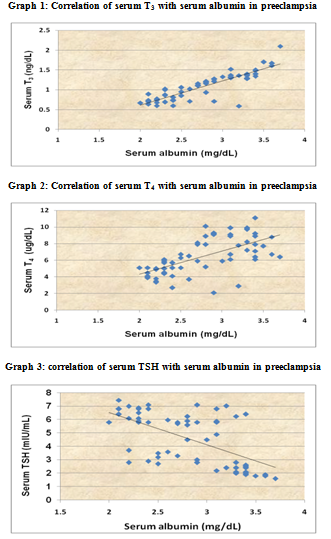
Results: We observed that out of 60 preeclampsia patients 13 (21.66%) women having hypothyroidism, 18 (30%) women having sub-hypothyroidism and 29 (48.33%) women were of euthyroid. The mean serum TSH in preeclamptic cases (preeclampsia women) were increased significantly (p >0.001). On the other hand serumT3 levels and Serum T4 levels were decreased significantly in preeclamptic women when compared to controls (p >0.001). Serum albumin was decreased significantly in preeclamptic women when as compared to controls (p >0.001). We found that there was statistically significant positive correlation between serum T3 and T4 and serum albumin (r = 0.8735, p = 0.0001) and statistically significant negative correlation between serum TSH and serum albumin (r = - 0.6109 p = 0.0001).
Conclusion: From the results all these parameters play an important role in preeclampsia hence early detection of these parameters in preeclampsia may help to improve maternal and fetal outcome.
Keywords: T3 -Total triiodothyronine, T4- Tetraiodothyronine, TSH- Thyroid stimulating hormone
Full Text:
Introduction
Pregnancy is a physiological state associated with many alterations in metabolic, biochemical, physiological, haematological and immunological processes. If these changes are exaggerated, they can lead to complications during pregnancy1.
Pregnancy induced hypertension presents with new onset hypertension and proteinuria after 20 weeks of gestation. Hypertensive disorders are the most common medical complications occurring during pregnancy. Pregnancy induced hypertension was reported with the incidence of about 10% of first pregnancy and 20% to 25% of previous pregnancy. The incidence of Pregnancy Induced Hypertension (PIH) varies among different hospitals, regions and countries.2
According to WHO's World Health Report 1998, Preeclampsia is defined as “the development of hypertension (>140/90 mm of Hg) after 20 weeks of pregnancy in a woman with proteinuria associated with or without edema having no previous history of hypertension"3
Recent finding related to pathophysiology of preeclampsia suggest that the endothelial cell injury and altered endothelial cell function play a pivotal role.4 It has been proposed5 that the poorly perfused placenta is the origin of a humoral factor that affects maternal systemic function, directly or indirectly, by activating endothelial cells, with the resultant vascular injury.5The characteristic pathologic lesion seen in the uteroplacental bed of patients with preeclampsia is a necrotizing arteriopathy consisting of fibrinoid necrosis, accumulation of foam cells or lipid?laden macrophages in the decidua, fibroblast proliferation and a perivascular infiltrate. This lesion has been termed "acute atherosis.6
Normal pregnancy is associated with physiological and hormonal changes. However abnormal hormonal changes especially thyroid hormone dysfunctions has also shown the relationship between developments of preeclampsia. High levels of TSH, low level of T3 and T4 were shown to be associated with development of preeclampsia and its complication.
In recent glomerular injury also has been considered as characteristic feature of preeclamptic women.7 More precisely it comprises endothelial cell swelling, obliteration of endothelial fenestrate leading to loss of proteins via proteinuria. Keeping all these facts in mind the present work was undertaken to evaluate the relationship of these parameters with preeclampsia.8,9
Serum T3, T4, TSH was measured by enzyme immunoassay on STAT FAX 4300 CHROMATE ELISA Reader using ERBA Thyroid kits by Aspen Laboratories Pvt. Ltd. The estimation of the serum albumin was done by BCG Dye binding method on fully automated XL300 analyse.
Material and methods
Hospital based cross sectional study was conducted in Department of Biochemistry with the help of Obstetrics and Gynaecology Department during period May 2013 to October 2014. The study was approved by institutional Ethics Committee for research work.
The study comprised of 60 cases of preeclamptic women who attended in Obstetrics and Gynaecology Department in Govt. Medical College, Nagpur from February 2013 to August 2014 and 60 normotensive healthy women as controls. These patients were all primigravidae aged between 18 to 30 years and in the third trimester of pregnancy
The diagnosis of Preeclampsia was done by the Department of Obstetrics and Gynaecology based on the definition given by American college of obstetrics and gynecology. Systolic blood pressure > 140 mm Hg or diastolic blood pressure >90 mm Hg; systolic blood pressure increase of > 30 mm Hg or diastolic blood pressure increase of 15 mm Hg over first trimester of pregnancy values (manifested on two occasion at least 6 hrs apart) and proteinuria ≥300 mg or greater in 24 hr urine collection or dipstick protein ≥ 1+ (on two occasion at least 6 hrs apart) is preeclampsia
The inclusion criteria for pre-eclamptic subjects include Age -18 to 30 years, primigravidae in the 3rd trimester of pregnancy, BP - 140/90 mm Hg in third trimester of pregnancy (ACOG criteria), Urine albumin ≥ | + dipstick or 300 mg per 24 hour urine. Normal pregnant women in third trimester of pregnancy were taken as controls.
The exclusion criteria for both the groups were Age < 18 years and > 30 years. (Multigravidae with more than one para, previous history of hypertension, diabetes mellitus, thyroid disorder, dyslipidemia, renal disease and convulsions, Family history of preeclampsia. After explaining all details, informed consent was taken from each subject for participation in this study. History of patient was recorded on preformed questionnaire which included detailed history about present pregnancy, family history of preeclampsia and exclusion criteria. 2 ml of fasting blood sample was collected in plane blub and serum is separated by centrifugation compare demographic, clinical and biochemical parameters between preeclampsia and control groups. Pearson’s correlation coefficient (r) was calculated to assess the correlation between serums TSH, T3 and T4 and serum albumin.
STATISTICAL ANALYSIS-
Unpaired t-test was performed to compare demographic, clinical and biochemical parameters between preeclampsia and control groups. Pearson’s correlation coefficient (r) was calculated to assess the correlation between serums TSH, T3 and T4 and serum albumin
Result
Characteristics in preeclampsia cases and normal pregnant controls
The mean age in the preeclampsia cases was 23.57 ± 2.99 years and that in controls was 22.90 ± 2.96 years. The mean gestational age of cases was 33.92 ± 2.42 weeks where as in controls 33.98 ± 2.48 weeks.(Table1) There was no significant difference in age and gestational age between cases and controls [p value > 0.05]. Study groups were well matched for age and gestational age.
In our study we found that out of 60 preeclampsia patients 13 (21.66%) women having hypothyroidism, 18 (30%) women having sub-hypothyroidism and 29 (48.33%) women were euthyroid. In normotensive pregnant controls all 60 women were euthyroid.
Comparison of serum thyroid profile
In the present study, in preeclamptic women the mean values of T3, T4 were found to be 1.10 ± 0.34 ng/mL and 6.54 ± 2.19 ug/dL respectively while in control group it was 1.47 ± 0.36 ng/mL and 8.46 ± 1.66 ug/dL. (Table 2) The results showed that mean T3 and T4 levels in preeclamptic women were significantly decreased as compared to control group with p value <0.0001. In preeclamptic women mean TSH level were found to be 4.49 ± 2.00 mlU/mL while in control group was found to be 2.95 ± 0.80 mlU/mL. The results showed that mean TSH levels in preeclamptic women were significantly increased as compared to control group with p value <0.0001.
The mean level of serum albumin in preeclampsia cases was 2.8 ± 0.49 mg/dL and in the normal pregnant women was 4.63 ± 0.37 mg/dL.(Table 2) There was low serum albumin in preeclampsia as compared to normal pregnant women and difference was statistically highly significant. (p=<0.001)
Serum T3 and T4 level correlated with serum albumin and r value was 0.8735 and 0.6299 respectively. This was statistically significant positive correlation. Serum TSH level correlated with serum albumin and r value was - 0.6109.This was statistically significant negative correlation. (p <0.001)
Discussion
Pregnancy is a physiological process. Pregnancy is associated with number of important physiological and hormonal changes.10Human chorionic gonadotrophin hormone secreted by the placenta stimulates thyroid gland during pregnancy due to its structural similarity with thyrotropin. Thyroid hormones have very important role in embryogenesis and fetal development.10,11 Normally pregnancy associated with mild hyperthyroidism on other hand preeclampsia associated with hypothyroidism.12,13 Normal pregnancy is state of hyperestrogenemia.14 During pregnancy increase in estrogens leads to increased thyroid binding globulin which is due to increase hepatic synthesis and increase the half life of thyroid binding globulin because of estrogen induced sialylation of protein.15 As a result of increase in thyroid binding globulin serum T3 and T4 level also increase. In addition there is increased production of type III deiodinase enzyme from placenta which may lead to increased production of the T3 and T4.15
Preeclampsia is a state of decreased estrogen, may be due to placental dysfunction. This decreased estrogen leads to decreased in synthesis of thyroid binding globulin.16,17 As thyroid binding globulin is decreased it might be the reason for lowering of serum T3 and T4 levels in preeclampsia.
Preeclampsia is pregnancy induced autotoxication multisystem disorder. In preeclampsia most affected organs are kidney, liver and brain. In liver and kidney peripheral conversion of the T4 to T3 occurs. Thus due to involvement of kidney and liver in preeclampsia no conversion of T4 to T3 occurs. This might be the main factor for decreased serum T3 concentration in preeclamptic patients.17,18 Preeclamptic women may have associated various condition like systemic illness, anorexia nervosa, starvation, protein energy malnutrition. This lead to reduced extra-thyroidal deiodination of T3 and T4. Change in thyroid profile in preeclampsia may be due to hypothalamic-pituitary dysfunction secondary to disease stress.18,19
The present finding of the our study resemble with studies of Basbug M et al,20 Khaliq F et al21, Pasupathi P et al17, Sardana D et al22, Khadem N et al18
The decrease TSH levels observed in our study is in accordance with the studies of Dhananjaya BS et al23,Kumar Ashok et al19, Das S et al16, Larijani B et al.24
Endothelium injury play important role in the pathogenesis of preeclampsia25. During the preeclampsia there may be endothelial damage. This may lead to endotheliosis and loss of endothelial fenestrations and disruption of the glomerular filtration barrier. Ultimately endothelial damage may lead to proteinuria.7
Various studies26,27 explained that decrease in the serum albumin during preeclampsia is due to proteinuria. Kidney eliminates waste product from the blood. This filtration occurs with help of the glomerular filtration barrier present in the Bowman’s capsule. Bowman’s capsule consists of three layers, first endothelial cell of the glomerular capillary, second a basement membrane, and third epithelial cell of bowman's capsules. The integrity of the glomerular filtration barrier avoids filtration of the protein. During the preeclampsia there is endothelial damage which may lead to endotheliosis and loss of endothelial fenestration and disruption of the glomerular filtration barrier which may lead to proteinuria.7
Serum T3 and T4 are mainly bound to thyroid binding globulin and also to some extent to pre-albumin and albumin.28 In preeclampsia there is loss of albumin in urine so hormones bound with to albumin are also lost in urine. This may lead low serum T3 and T4 levels. This suggests a strong relationship between loss of albumin with low serum T3 and T4 levels in preeclamptic women.13,22,29
As per our results the correlation between TSH and albumin appear to be is significantly negative. It might be due to fact that the pre-eclamptic patients are in a state of mild hypothyroidism. Lao TT et al30, Tolino A et al31 Farah Khaliq et al21 also found significant negative correlation between serum TSH and serum albumin.
Conclusion
Our results suggest that diseases of the thyroid gland itself might be a predisposing factor for the development of preeclampsia.
In addition to this serum albumin was decreased in preeclamptic women as compared to normal pregnant women. Hence at this juncture it can be suggested that serum albumin could be used as predictor of severity of preeclampsia.
A positive correlation in serum albumin was observed with T3 and T4 and negatively correlated with serum TSH level in preeclamptic women.
As study was conducted on small group further study with large group is needed for confirmation of our finding.
Acknowledgement
We express our deep gratitude to all patients for their cooperation. We acknowledge the immense help received from the scholars whose articles are cited and included in references of this manuscript. I am thankful to Dr. Muddershwar Head Biochemistry of the department Govt. Medical College, Nagpur. Also we are thankful to Dr. Manisha Achawale for their guidance. Authors acknowledge the immense help received from the scholars whose articles are cited and included in references of this manuscript. The authors are also grateful to authors/editors/publishers of all those articles, journals and books from where the literature for this article has been reviewed and discussed.
Source of funding: Nil
Conflict of interest: Nil
References:
1. Soma-Pillay P, Nelson-Piercy C; Tolppanen H, Mebazaa A. Physiological changes in pregnancy. Cardiovasc J Afr. 2016;27(2):89-94.
2. Ghosh MK. Maternal Mortality: A Global Perspective. J Reprod Med. 2001;46(5):427-33.
3. World Health Organization. The World Health Report 1998 . Life in the 21st century. 1998. p. 97.
4. Roberts J, Redman C. Pre-eclampsia: more than pregnancy-induced hypertension. Lancet. 1993 Jun;341(8858):1447-51.
5. Rodgers GM, Taylor RN, Roberts JM. Preeclampsia is associated with a serum factor cytotoxic to human endothelial cells. Am J Obstet Gynecol. Elsevier; 1988 Oct 10;159(4):908-14.
6. Goode GK, Miller JP, Heagerty AM. Hyperlipidaemia, hypertension, and coronary heart disease. Lancet. 1995 Feb;345(8946):362-4.
7. Kuang-Yu J, Laszik ZG. Renal Effects of Preeclampsia [Internet]. Available from: www.intechopen.com
8. Stillman IE, Karumanch SA. The Glomerular Injury of Preeclampsia. J Am scocity Nephrol. 2007;18:2281-4.
9. Strevens H, Wide-Swensson D, Hansen A, Horn T, Ingemarsson I, Larsen S, et al. Glomerular endotheliosis in normal pregnancy and pre-eclampsia. An Int J Obstet Gynaecol. 2003 Sep;110(9):831-6.
10. Pasupathi P, Chandrasekar V, Kumar US. Thyroid Hormone Changes in Pregnant and Non-Pregnant Women: A Case-Control Study. Thyroid Sci. 2009;4(3):3-7.
11. Khandakar M, Ali AR, Kahtun M. Thyroid status of normal pregnant women in Dhaka City. Mymensingh Med J. 2002;11(1):1- 5.
12. Qublan HS, Al-Kaisi IJ, Hindawi IM, Hiasat MS, Awamleh I, Hamaideh a. H, et al. Severe pre-eclampsia and maternal thyroid function. J Obstet Gynaecol. 2003 Jan;23(3):244-6.
13. Kaya E, Sahin Y, Ozkceci Z, Pasaoglu H. Relation between birth weight and thyroid function in preeclampsia. Gynecol Obstet Invest. 1994;37(1):30-3.
14. Hubel CA. Oxidative stress in the pathogenesis of preeclampsia. Proc Soc Exp Biol Med. 1999;222(3):222–35.
15. Fantz CR, Dagogo-jack S, Ladenson JH, Gronowski AM. Thyroid Function during Pregnancy. Clin Chem. 1999;45(12):2250- 8.
16. Das S, Char D, Sarkar S, Saha TK, Biswas S. Evaluation of Thyroid Hormone Changes in Non-Pregnant , Normotensive Pregnant and Pregnancy with Preeclampsia. J Dent Med Sci. 2013;11(6):16-8.
17. Pasupathi P, Deepa M, Rani P, Vidhya Sankar K, SP satish kumar. Evaluation of Serum Lipids and Thyroid Hormone Changes in Non-Pregnant, Pregnant, and Preeclampsia Women. Thyroid Sci. 2009;4(10):2-7.
18. Khadem N, Ayatollahi H, Roodsari FV, Ayati S, Dalili E, Shahabian M, et al. Comparison of serum levels of Triiodothyronine ( T3 ), Thyroxine ( T4 ), and Thyroid Stimulating Hormone ( TSH ) in preeclampsia and normal pregnancy. Iran J Reprod Med. 2012;10(1):47-52.
19. Kumar A, Ghosh BK, Murthy NS. Maternal thyroid hormonal status in preeclampsia. Indian J Med Sci. 2005;59(2):57-63.
20. Basbug M, Aygen E, Tayyar M, Tutus A, Kaya E, Oktem O. Correlation between maternal thyroid function tests and endothelin in preeclampsia-eclampsia. Obstet Gynecol. 1999;94(4):551-5.
21. Khaliq F, Singhal U, Arshad Z, Hossain MM. Thyroid functions in pre-eclampsia and its correlation with maternal age, parity, severity of blood pressure and serum albumin. Indian J Physiol Pharmacol. 1999;43(2):193-8.
22. Sardana D, Nanda S, Kharb S. Thyroid hormones in pregnancy and preeclampsia. J Turkish Ger Gynecol Assoc. 2009;10(3):168-71.
23. Dhananjaya B, Kumaran DS, Venkatesh G, Murthy N, Shashiraj H. Thyroid Stimulating Hormone (TSH) Level as a Possible Indicator of Pre-eclampsia. J Clin Diagnostic Res. 2011;5(8):1542-3.
24. Larijani B, Marsoosi V, Aghakhani S, Moradi A, Hashemipour S. Thyroid hormone alteration in pre-eclamptic women. Gynecol Endocrinol. 2004;18(2):97-100.
25. Roberts JM, Taylor RN, Musci TJ, Rodgers GM, Hubel CA, McLaughlin MK. Preeclampsia: An endothelial cell disorder. Am J Obstet Gynecol. Elsevier; 1989 Nov 11;161(5):1200–4.
26. Muzammil S, Khayyam UK, Siddiqui N ali. Serum protein ratio in normal and pre-eclamptic women of primiparous and multiparous in relation to age. Int J Basic Appl Med Sci. 2014;4(2):331- 5.
27. Olooto W, Amballi A. Assessment of Total Protein, Albumin, Creatinine and Aspartate Transaminase level in Toxemia of Pregnancy. J Med …. 2013;12(8):791-6.
28. Demers L, Spencer C. The Thyroid: pathophysiology and thyroid functioning testing. In: Tietz textbook of clinical chemistry and molecular diagnostics. 4th ed. Elsevier; 2006. p. 2053-96.
29. Osathanondh R, Tulchinsky D, Chopra IJ. Total and free thyroxine and triiodothyronine in normal and complicated pregnancy. J Clin Endocrinol Metab. 1976;42(1):98-104.
30. Lao TT, Chin RK, Swaminathan R, Lam YM. Maternal thyroid hormones and outcome of preeclmaptic pregnancies. An Int J Obstet Gynaecol. 1990;97(1):71-4.
31. Tolino A, de Conciliis B, Montemagno U. Thyroid hormones in the human pregnancy. Acta Obstet Gynecol Scand. 1985;64(7):557-9.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License