IJCRR - 5(2), January, 2013
Pages: 08-19
Print Article
Download XML Download PDF
PHYTOCHEMISTRY, ANTIOXIDANT AND ANTIBACTERIAL ACTIVITIES OF MEDICINAL PLANTS- A COMPARATIVE STUDY
Author: Rajamurugan R., Deepa V., Sivashanmugam M., Raghavan C. M.
Category: General Sciences
Abstract:Plants are the most important sources of medicines. Today the large number of drugs in use is derived from plants. The important advantages for therapeutic uses of medicinal plants in various ailments are their safety besides being economical, effective and easy availability. The present investigation was aimed to screen phytochemicals, antioxidant and antibacterial activities of ethanolic extracts of leaves of Aegle marmelos, Ocimum sanctum, Alternanthera sessilis, Eclipta alba, and Acalypha indica. The phytochemical analysis revealed the presence of terpenoids, phenols, flavonoids, tannins, alkaloids, cardiac glycosides, and steroids in all the extracts. The antioxidant activity of the extracts was evaluated by DPPH, ABTS and reducing power assay. All the extracts showed significant antioxidant activity. The plant preparations were also screened individually for antibacterial activity against three selected bacterial sp by agar-well diffusion method. Results showed that all the extracts were effective against bacterial sp tested.
Keywords: Aegle marmelos, Ocimum sanctum, Alternanthera sessilis, Antioxidant activity, Antimicrobial activity.
Full Text:
INTRODUCTION
Current advancements in drug discovery technology and search for novel chemical diversity have intensified the efforts for exploring leads from Ayurveda, the traditional system of medicine in India. Ayurvedic system of medicine has its long history of therapeutic potential. The use of plant extracts and phytochemicals both with known antimicrobial properties is of great significance, in the past few years several investigations have been conducted worldwide to prove antioxidant and antimicrobial activities from medicinal plants (Alonso-Paz et al., 1995; Nascimento et al., 1990). For a long period of time, plants have been a valuable source of natural products for maintaining human health, especially in the last decade, with more intensive studies for natural therapies (Sukanya et al., 2009). The medicinal plants are rich in secondary metabolites which are potential sources of drugs (Nadeem et al., 2010). It has been reported the free radical scavenging and antimicrobial activity of many medicinal plants are responsible for therapeutic effects against cancer, inflammatory, cardiovascular and infectious diseases. Since the use of medicinal plants have seconded a wide range in treating diseases, the present study concentrates on antioxidant and antibacterial activities of commonly available medicinal plants Aegle marmelos, Ocimum sanctum, Alternanthera sessilis, Eclipta alba, and Acalypha indica. Aegle marmelos (L) (Rutaceae) is growing wild throughout deciduous forest of India. Its fruits and leaves are valued in indigenous medicine (Charakbraty et al., 1960). The plant has been employed for long time in folk therapy. Poultice made of leaves are used for ophthalmia and ulcers. The leaves are use to lowering the blood glucose levels (Ayurvedic Pharmacopoeia of India, 1988). Other actions like antifungal (Renu, 1983), antibacterial (Banerji and Kumar, 1980), antiprotozoal (Banerjee, 1980), antispermatogenic (Sur et al, 1999) are also reported. Ocimum sanctum Linn. (Labiatae), commonly known as holy basil, is an herbaceous plant found throughout the South Asian region (Hannan et al 2007). The oil of Ocimum sanctum possesses antibacterial, antifungal, antioxidant and radioprotective properties (Sharma et al., 2002). Ancient Hindu literature is rich with the medical actions of Ocimum sanctum (Ubiad et al., 2002). Alternanthera sessilis is a prostrate or perennial herb. The branches are raised from the root and are up to 50 cm long. This plant is found in damp places, wet headlands, roadsides and sometimes as weed of plantations. In South East Asia this plant is taken as vegetables. In India it is used for treatment of gastrointestinal problems (Archana et al., 2011). Eclipta alba L. Hassk. (Asteraceae) has been reported for treating liver was cirrhosis and infective hepatitis (Chopra et al., 1966). In Ayurveda, the root powder is used for viz. treating hepatitis, enlarged spleen and skin disorders. Mixed with a little oil when applied to the head, the herb relieves headache. The extract of its leaves is mixed honey and given to infants, for to expel worms. Eclipta alba is also given to children in case of urinary tract infections (Prabu et al., 2011). Acalypha indica belongs to the family Euphorbiaceae. The leaf can be used for the treatment of throat infections and wound healing, and also used as anti-venom and migraine pain relief. (Oudhia, 2003; Valsara, 1994).
MATERIALS AND METHODS
Extraction of plant material
The plant materials (Aegle marmelos (AM), Ocimum sanctum (OS), Alternanthera sessilis (AS) Eclipta alba (EA), and Acalypha indica (AI)) were collected locally. Authentication of the plant materials was made by Dr.M.Shivashanmugam, Asst professor in Botany, M.G.R College, Hosur. Leaves were detached and dried in shade. About 100 gms of dried leaves were ground to powder and exhaustively extracted with 600 ml ethanol separately using soxhlet apparatus and extracts were concentrated under reduced pressure and then stored in an air tight container for further study.
Phytochemical screening
Phytochemical screening was performed using standard procedures (Sofowora et al., 1993; Trease, 1989).
Evaluation of antioxidant activity
DPPH radical scavenging assay The free radical scavenging activity of all the extracts was measured by decrease in the absorbance of ethanolic solution of DPPH (Braca et al., 2001). Different concentration of extracts (10-500 µg/ml) and positive control ascorbic acid was added to 2 ml of freshly prepared DPPH. The measurement was performed in triplicates. After incubation for about 30 min at room temperature in the dark, the absorbance was measured at 520 nm using spectrophotometer (RAYLEIGH).
Radical scavenging activity (%) was calculated using the following formula:-
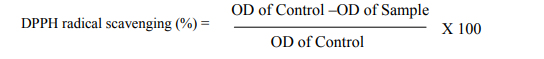
ABTS radical cation-scavenging activity
The antioxidant activities of extracts were determined by the improved ABTS. + radical cation scavenging ability as described by Baltrusaityte et al (2007) with minor modifications. ABTS + radical cation was produced by mixing 7 mM 2,2'- azino-bis (3- ethylbenzothiozoline-6-sulphonic acid) diammonium salt (ABTS) and 2.45 mM potassium persulfate (K2S2O8), incubated at room temperature in dark. To determine the ABTS radical scavenging activity, 3.9 ml of ABTS+ solution was mixed thoroughly with 0.1 ml of different concentration (10-500 µg/ml) of extracts. The reaction mixture was allowed to stand at room temperature for 6 min and the absorbance was immediately measured at 734 nm. Appropriate blank was prepared and the percentage decrease in absorbance was calculated by the following formula:

Reducing power assay
The reducing power of the extracts determined as per Oyaizu et al. (1986) method. Different concentrations of the extract (10-500 µg/ ml) in 1ml of ethanol were mixed with phosphate buffer (2.5 ml, 0.2M, pH 6.6) and potassium ferrocyanide (2.5 ml, 1%). The mixture was incubated at 50 C for 20 min. To a portion (2.5 ml) of the reaction mixture, trichloroacetic acid (10%) was added, which was then centrifuged at 3000 rpm for 10 minute upper layer of the solution (2.5 ml) was mixed with distilled water (2.5 ml) and ferric chloride (0.5ml, 0.1%) and the absorbance was measured at 700 nm and compared with reference standard ascorbic acid. Increase in absorbance of the reaction mixture indicates reducing power of the sample extracts.
Determination of antimicrobial activity
Test microbial cultures Test bacterial cultures such as Escherichia coli (E.coli), Staphylococcus aureus (S. aureus), and Proteus mirabilis (Pr. mirabilis) were obtained from Post Graduate and research department of Microbiology, M.G.R College, Hosur. Test organisms were sub cultured periodically and maintained on their respective growth media for further study. temperature in dark. To determine the ABTS radical scavenging activity, 3.9 ml of ABTS+ solution was mixed thoroughly with 0.1 ml of different concentration (10-500 µg/ml) of extracts. The reaction mixture was allowed to stand at room temperature for 6 min and the absorbance was immediately measured at 734 nm. Appropriate blank was prepared and the percentage decrease in absorbance was calculated by the following formula:

Screening of antimicrobial activity
The modified (Collins et al., 1995) agar-well diffusion method was employed to determine the antimicrobial activities of all the leaf extracts against three bacterial strains E. coli, Pr. mirabilis, and S. aureus. The bacterial cultures were inoculated into Muller Hinton agar and incubated at 37 ?C. Approximately, 10ml of sterile Muller Hinton agar was poured in to sterile culture plates and allowed to set wells of about 8 mm in diameter were punched on the plates. About 25-500 μg/ml of the extracts were dispensed in to the wells and the plates were incubated at 37 ?C and observed after 24 h.
Statistical analysis
Results are expressed as the mean ± S.D. of three independent experiments (n=3). Student’s t-test was used for statistical analysis; P values > 0.05 were considered to be significant. IC50 was calculated by linear regression analysis using Graph pad prism statistical software.
RESULTS AND DISCUSSION
Phytochemical screening
Presence of bioactive constituents such as phenols, terpenoids, tannins, flavonoids, and saponins in the crude extracts of plant origin contribute to the antimicrobial, antifungal and DPPH radical scavenging activity Free radicals have been a subject of significant interest among scientists in the past decade and their possible role in human diseases has gained importance nowadays (Maxwell, 1995; Jovanovic and Simic, 2000). Antioxidants neutralize free radicals that are defined as atoms or groups of atoms having an unpaired electron (Finkel and Holbrook, 2000). These also include related reactive oxygen species (ROS) that leads to free radical generation, causes the cascading chain reaction in biological system. Antioxidants present in various dietary supplements offered their beneficial effects by neutralizing these ROS during various disease conditions. DPPH radical is widely used as a model to investigate the scavenging potential of several natural compounds such as phenolics or crude extract of plants (Veerapur et al., 2009). In the present study, the ethanolic extracts of Aegle marmelos, Ocimum sanctum, Alternanthera sessilis, Eclipta alba, and Acalypha indica exhibited marked DPPH radical scavenging activity significantly. Lower absorbance of the reaction mixture indicates higher the free radical scavenging activity. Fig. 1 shows that % inhibition decreases the concentration of DPPH radical due to scavenging ability of standard ascorbic acid, as a reference compound and that of the ethanolic extracts. The scavenging effect of AM, OS, AS, EA, and AI and standard ascorbic acid on the DPPH radical decreased in the order: ascorbic acid > AM > AI > OS > EA >AS. The IC50 values for these compounds were found to be 92 μg/ml, 98 μg/ml, 154 μg/ml, 178 μg/ml, 324 μg/ml and 522 μg/ml respectively (Table 3). The results also show that the ethanolic extract of AM possesses significantly higher DPPH scavenging activity than other extracts and were most potent among the five extracts.
ABTS radical scavenging activity
The ABTS assay is based on the inhibition of the radical cation ABTS+ , which has a characteristic long wavelength absorption spectrum (SanchezMoreno, 2002). The ABTS chemistry involves direct generation of ABTS radical mono cation with no involvement of any intermediary radical, it is a decolorization assay, thus the radical cation is formed prior to addition of antioxidant test system rather than the generation of radical taking place continually in the presence of antioxidant. This method is used for the screening of antioxidant activity of herbal preparations, is applicable to both lipophilic and hydrophilic antioxidants (Long et al., 2000). In this study, ABTS radical was effectively scavenged by the ethanloic extract of AM, followed by AI, OS, EA, and AS in a concentration dependent manner but the scavenging effect was lesser than that of the reference compound ascorbic acid (Fig.2). IC50 values were 102 μg/ ml, 110 μg/ ml, 252 μg/ ml, 288 μg/ ml, and 364 μg/ ml for AM, AI, OS, EA, and AS respectively. Similarly IC50 for standard was 96 μg/ ml (Table 4).
Reductive ability
The reducing capacity of a compound may serve as a significant indicator of its potential antioxidant activity (Blazovics et al, 2003). Reducing power is to the measure of the reductive ability of antioxidant and it is evaluated by the transformation of Fe3+ to Fe2+ in the presence of sample extracts (Gulcin et al., 2003).
In this study, the reducing capacities of the extracts were also determined. Results of the reducing power of standard antioxidant ascorbic acid and the ethanolic extracts of AM, OS, AS, EA and AI are shown in Fig. 3. The data show an increase in the reducing power of the extracts with increase in dosage. However, the ethanolic extract of OS presents better activity at a concentration of 500 µg/ml followed by EA, AS, AM, and AI (Table 5).
Antibacterial activity
Plant based antimicrobial compounds have enormous therapeutical potential as they can serve the purpose without any side effects that are often associated with synthetic antimicrobials. The active ingredients of the plant parts are better extracted with alcohol than other solvents. The alcohol extracts contain alkaloids, coumarins and tannins (Okemo, 1996). Coumarins and tannins have antibacterial and antihelminthic properties (Hedberg et al., 1983). Eloff (1998) and Cowan (1999) found that alcohol was more efficient than acetone in extracting phytochemicals from plant materials. In the present work antibacterial effect of leaf extracts from five plants were studied on bacterial cultures (two gram negative bacteria, Proteus mirabilis and Escherichia coli and one gram positive bacterium Staphylococcus aureus). Results of this study revealed that the ethanolic extracts exhibited better antibacterial activity towards all bacterial isolates tested. There was a significant variation in the antibacterial activities of the leaf extracts. The ethanolic extract of AM exhibited highest antibacterial activity followed by OS, AS, EA and AI with diameter of inhibition zone values between 5 and 26 mm. Maximum inhibitory effect was recorded for E. coli 26 mm by AM; followed by Pr. mirabilis 25 mm by OS; for S. aureus, 24 mm by AS (Table 6 and Figures 4, 5, 6 and 7). Tannins were found to be excellent antibacterial compounds (Cowan, 1999). They have diverse effects on biological systems because they are potential metal ion chelators, protein precipitating agents and biological antioxidants (Hagerman, 2002). A number of mechanisms have been proposed to explain the tannin’s antimicrobial activity which includes extracellular microbial enzymes and proteins, deprivation of iron as substances for microbial growth or direct action towards its membranes (Scalbert, 1991). Condensed tannins have been determined to bind cell walls of luminal bacteria, preventing growth and protease activity (Jones et al., 1994). From the results of this investigation, it is therefore postulated that tannins present in the crude extracts were responsible for the antibacterial activity.
CONCLUSION
From the results of this study it could be concluded that all the five medicinal plants contain bioactive principles and found to have a strong antioxidant and antibacterial activities specifically in the ethanolic extract of leaves. Results of our findings further confirm the use of these herbs as traditional medicine and may be used as effective and potential sources of novel antioxidant and antimicrobial drugs
References:
1. Alonso-Paz E, Cerdeiras MP, Fernandez J, Ferreira F, Moyna P, Soubes M, Vazquez A, Veros S, Zunno L (1995). Screening of Uruguayan medicinal plants for antimicrobial activity. J. Ethanopharm. 45: 67-70.
2. Nascimento S C, Chiappeta A, Lima R M (1990). Antimicrobial and cytotoxic activities in plants from pernambuco, Braz. Fitoter. 61: 353- 355.
3. Sukanya S L, Sudisha J, Hariprasad P, Niranjana S R, Prakash H S and Fathima S K (2009). Antimicrobial activity of leaf extracts of Indian medicinal plants against clinical and phytopathogenic bacteria. African Journal of Biotechnology, 8 (23): 6677-6682.
4. Nadeem Ahmad Siddique, Mohd Mujeeb, Abdul Kalam Najmi and Mohd Akram (2010). Evaluation of antioxidant activity,quantitative estimation of phenols and flavonoids in different parts of Aegle marmelos. African Journal of Plant Science, 4 (1): 001-005
5. Charakbraty B, Malik C, Bhatthacharya S (1960). Studies on the effect of green leaves of Aegle marmelos and Piper nigrum on the glucose and cholesterol levels of blood in diabetes mellitus. Indian Med. Forum 9: 285- 28.
6. Ayurvedic Pharmacopoeia of India (1988). The Ayurvedic Pharmacopoeia of India. VolIV, Part- I, Ist -Edition, Govt. of India, Ministry of health and family welfare, Dept. of AYUSH, New Delhi.
7. Renu (1983). Fungitoxicity of leaf extracts of some higher plants against Rhizoctonia solani Kuehn. Natl. Acad. Sci. Lett. 6: 245-246
8. Banerji N and Kumar R (1980) Studies on the seed oil of A. marmelos and its effect on some bacterial species. J. Inst. Chem., 52.
9. Banerjee A (1980). Antimicrobial and anti helminthic screening of the fixed oil and unsaponefiable matter of Aegle marmelos. Conf. Proc. 67th session, Indian Science Congress. Calcutta Part III. Chemistry Section, Abstr., 247.
10. Sur T K, Pandit S, Pramanik T (1999). Antispermatogenic activity of leaves of Aegle marmelos, corr. In albino rats: a preliminary report. Biomed. 19:199-202.
11. Hannan J M A, Marenah L, Ali L, Rokeya B, Flatt P R, and Abdel-wahab Y H A (2006). Ocimum sanctum leaf extracts stimulate insulin secretion from perfused pancreas, isolated islets and clonal pancreatic β-cells. J Endocrinol. 189:127–136.
12. Sharma M K, Kumar M, and Kumar A (2002). Ocimum sanctum aqueous leaf extract provides protection against mercury induced toxicity in Swiss albino mice. Ind J Exp Biol. 40:1079–1082.
13. Ubaid F S, Anantrao K M, Jaju J B, Mateenuddin M D (2002). Effect of Ocimum sanctum leaf extract on hepatotoxicity induced by antitubercular drugs in rats. Ind J Phy Pharm. 47(4):465–470.
14. Archana Borah, Yadav R N S and Unni B G (2011). In vitro antioxidant and free radical scavenging activity of Alternanthera sessilis. International journal of pharmaceutical sciences and research, 2(6): 1502-1506
15. Chopra R N, Nayar S L, and Chopra I C (1966).Glossary of Indian Medicinal Plants. New Delhi: Council of Scientific and Industrial Res., pp: 104.
16. Prabu K, Shankarlal S, Natarajan E, and Mohamed Sadiq A (2011). Antimicrobial and Antioxidant Activity of Methanolic Extract of Eclipta alba. Advances in Biological Research, 5 (5): 237-240.
17. Oudhia P (2003). Traditional medicinal uses in India. J. Planta Medica. 15(5): 175-179.
18. Valsara T (1994). Screening of Indian Medicinal plants for antimicrobial activity. J. Ethnobiol. 35(3): 275-283
19. Sofowora A (2006). Medicinal Plants and Traditional Medicine in Africa.2nd edn., Spectrum Books Ltd., Ibadan, Nigeria, pp 151-153, 209-214.
20. Trease G E, and Evans W C (2002). Pharmacognosy. 15th ed. Saunders, London. pp. 53-336.
21. Braca A, De Tommasi N, Di Bari L, Pizza C, Politi M, and Morelli I (2001). Antioxidant principles from Bauhinia tarapotensis. J Nat Prod. 64: 892–895.
22. Baltrusaityte V, Venskutonis PR, and Ceksteryte V (2007). Radical scavenging activity of different floral origin honey and beebread phenolic extracts. Food Chem. 101: 502–514.
23. Oyaizu M (1986). Studies on products of browning reaction prepared from glucosamine. Jpn. J. Nutr. 44:307-314.
24. Collins C H, Lynes P M and Grange J M (1995). Microbiological methods, 7th edn.Butterwort-Heinemann Ltd. Britain, pp 175- 190
25. Ahmad I and Beg A Z (2001). Antimicrobial and phytochemical studies on 45 Indian medicinal plants against multi-drug resistant human pathogens. J. Ethnopharmacology, 74: 113-23.
26. Chapagain B P, Wiesman Z, and Tsror L (2007). In vitro study of the antifungal activity of saponin-rich extracts against prevalent phytopathogenic fungi. Industrial Crops and Products, 26:109-15.
27. Xiong Hai-Rong, Luo J, Hou W, Xiao H, and Yang Zhan-Qiu, (2011). The effect of emodin, an anthraquinone derivative extracted from the roots of Rheum tanguticum, against herpes simplex virus in vitro and in vivo. J. Ethnopharmacology, 133: 718- 723.
28. Maxwell S R (1995). Prospects for the use of antioxidant therapies. Drugs 49: 345-361.
29. Jovanovic S V and Simic M G (2000). Antioxidants in nutrition. Ann. N. Y. Acad. Sci. 899:326-634. 30. Finkel T and Holbrook N J (2000). Oxidants, oxidative stress and the biology of ageing. Nature, 408: 239-247.
31. Veerapur V P, Prabhakar K R, Parihar V K, Kandadi M R, Ramakrishana S, Mishra B, Satish Rao S B, Srinivasan K K, Priyadarsini K I, and Unnikrishnan M K (2009). Ficus racemosa stem bark extract: a potent antioxidant and a probable natural radioprotector. Evid. Based Complement Alternat. Med. 6: 317-324.
32. Sanchez-Moreno C (2002). Methods used to evaluate the free radical scavenging activity in foods and biological systems. Food Sci Tech Int. 8: 121-137.
33. Long L H, Kwee D C, and Halliwell B (2000). The antioxidant activities of seasonings used in Asian cooking. Powerful antioxidant activity of dark soy sauce revealed using the ABTS assay. Free Radic Res. 32: 181-186.
34. Blazovics A, Lugasi A, Szentmihalyi K, and Kery A (2003). Reducing power of the natural polyphenols of Sempervivum tectorum in vitro and in vivo: Acta Biological Szegediensis, 47 (1?4):99? 102.
35. Gulcin I, Oktay M, Kirecci E, and Kufrevioglu I (2003). Screening of antioxidant and antimicrobial activities of anise (Pimpella anisum L.) Seed extracts: Food Chem 83:371?382.
36. Okemo P O (1996). Antimicrobial efficacy of selected medicinal plants used by Kenyan Herbal doctors. Ph.D. thesis, Kenyatta University of Nairobi, pp. 173-90
. 37. Hedberg I, Hedberg O, Madati P, Mshigeni KE, Mshiu EN, and Samuelsson G (1983). Inventory of plants used in traditional medicine in Tanzania. II. Plants of the family Dilleniaceae to Opiliaceae. J. Ethnopharm. 9: 105-128.
38. Eloff J N (1998). Which extract should be used for screening and isolation of antimicrobial components from plants. J. Ethnopharm. 60: 1-8.
39. Cowan M M (1999). Plant products as antimicrobial agents. Clin. Microbiol. Rev. 12: 564-582.
40. Cowan M M (1999). Plants products as antimicrobial agents. Clin. Microbiol. Rev. 12: 564-582.
41. Hagerman A E (2002). Tannin chemistry. Department of Chemistry and Biochemistry, Miami University, USA.
42. Scalbert A (1991). Antimicrobial properties of tannin. Phytochem. 30:3875-3883.
43. (Jones G A, McALLISTER T A, MUIR A D, CHENG K J (1994). Effects of saifoin (Onobrychis viciifolia Scop.) condensed tannins on growth and proteolysis by four strains of luminal bacteria. Appl. Environ. Microbiol. 60: 1374-1378.
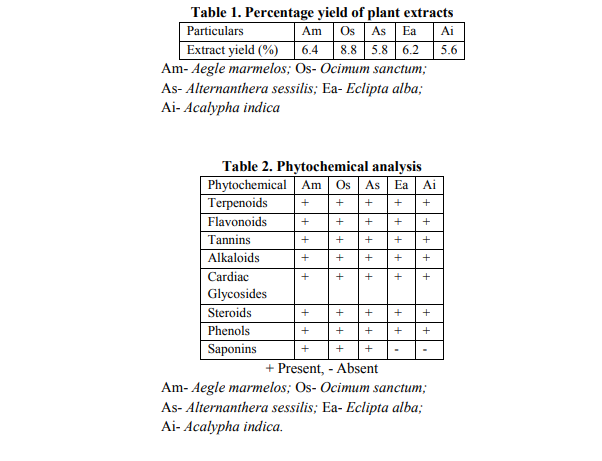
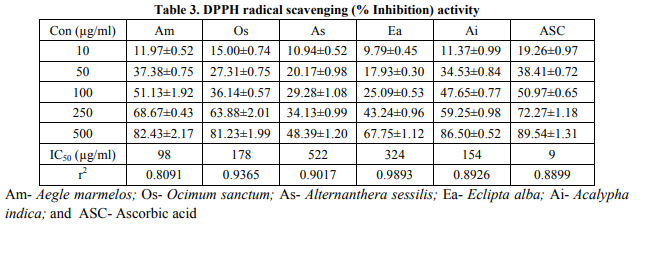
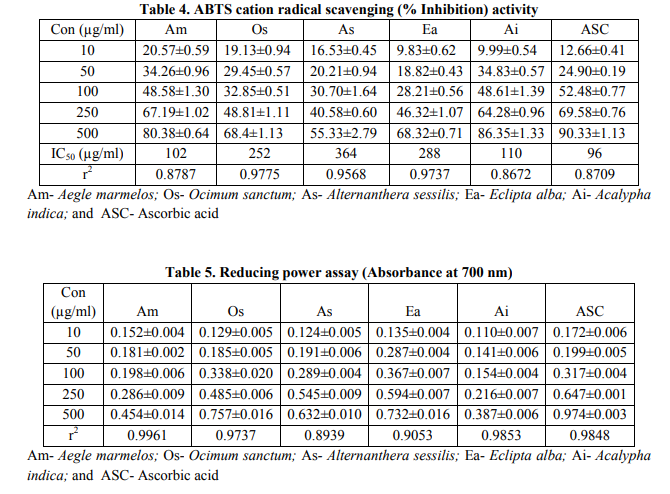

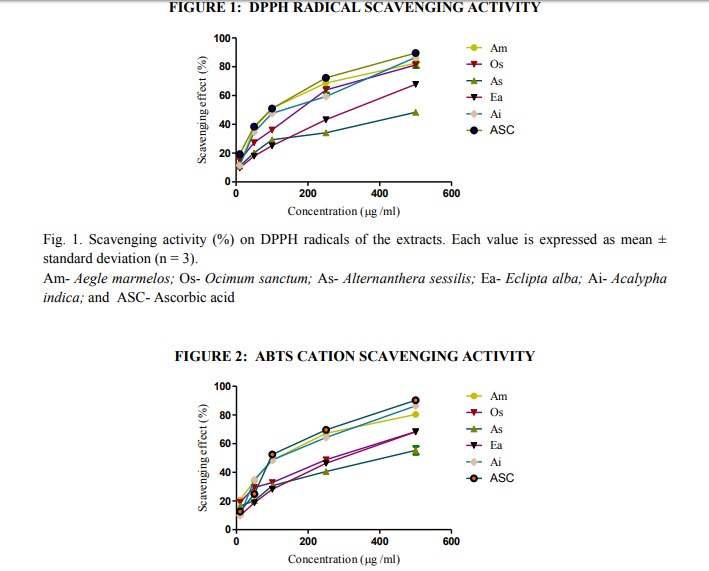
Fig. 2. Scavenging activity (%) on ABTS radicals of the extracts. Each value is expressed as mean ± standard deviation (n = 3). Am- Aegle marmelos, Os- Ocimum sanctum, As- Alternanthera sessilis, Ea- Eclipta alba, and AiAcalypha indica; and ASC- Ascorbic acid
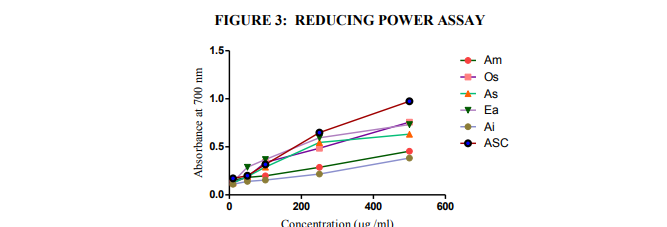
Fig. 3. Reducing power of the extracts (higher absorbance indicates higher reducing power). Each value is expressed as mean ± standard deviation (n = 3). Am- Aegle marmelos, Os- Ocimum sanctum, As- Alternanthera sessilis, Ea- Eclipta alba, and AiAcalypha indica; and ASC- Ascorbic acid
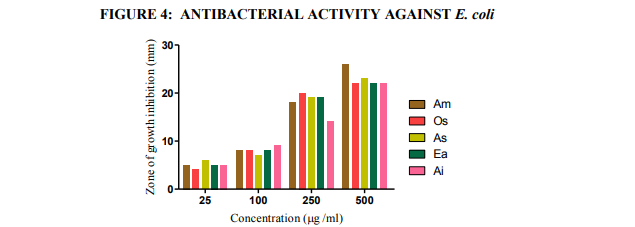
Fig.4. Antibacterial activities of the extracts against E. coli (agar-well diffusion method). Am- Aegle marmelos, Os- Ocimum sanctum, As- Alternanthera sessilis, Ea- Eclipta alba, and Ai- Acalypha indica
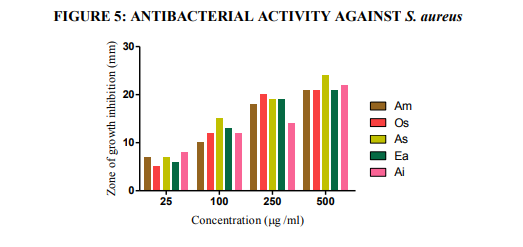
Fig.5. Antibacterial activities of the extracts against S. aureus (agar-well diffusion method). Am- Aegle marmelos, Os- Ocimum sanctum, As- Alternanthera sessilis, Ea- Eclipta alba, and Ai- Acalypha indica

Fig.6. Antibacterial activities of the extracts against Pr. mirabilis (agar-well diffusion method). AmAegle marmelos, Os- Ocimum sanctum, As- Alternanthera sessilis, Ea- Eclipta alba, and Ai- Acalypha indica.
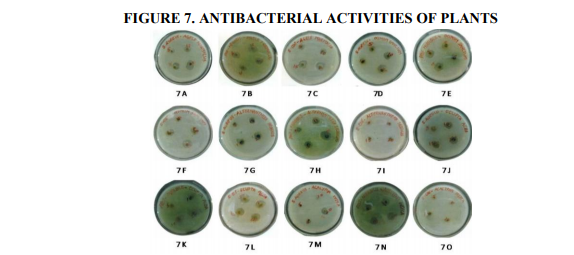
Fig.7. Antibacterial activities of the extracts (agar-well diffusion method). 7 A- Aegle marmelos, S.aureus; 7 B- Aegle marmelos Pr. mirabilis; 7 C- Aegle marmelos, E. coli; 7 D- Ocimum sanctum, S.aureus; 7 E- Ocimum sanctum, Pr. mirabilis; 7 F- Ocimum sanctum, E. coli; 7 G- Alternanthera sessilis, S.aureus; 7 H- Alternanthera sessilis, Pr. mirabilis; 7 I- Alternanthera sessilis, E. coli; 7 JEclipta alba, S.aureus; 7 K- Eclipta alba, Pr. mirabilis; 7 L- Eclipta alba, E. coli; 7 M- Acalypha indica, S.aureus; and 7 N- Acalypha indica, Pr. mirabilis;7 O- Acalypha indica, E. coli.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License