IJCRR - 5(4), February, 2013
Pages: 114-120
Date of Publication: 28-Feb-2013
Print Article
Download XML Download PDF
A STUDY OF SERUM CA-125 AND SALIVARY AMYLASE IN OVARIAN NEOPLASM IN A TERTIARY CARE HOSPITAL OF KOLKATA
Author: Sukla Nath, Swati Bhattacharyya, Rituparna Maji, H.N. Das , Shikha Das , Ranadip Chowdhury
Category: Healthcare
Abstract:Introduction: Ovarian cancer is the fifth most lethal malignancy in women in the United States and second most common of all genital cancers in developing countries including India .Ovarian cancer is curable in early stages, and seldom curable in advanced stages. Objective: Study was conducted to determine (1) whether pre operative serum level of CA-125 could differentiate between Benign and Malignant neoplasm (2) to evaluate the significance of serum salivary amylase in ovarian neoplasm. Material and Methods: This was a cross sectional descriptive observational study conducted in a tertiary care hospital in north Kolkata, West Bengal, India. Detailed information regarding associated diseases and investigation reports were recorded in a case record form. Results: Serum CA-125 was analysed with pre-operative serum sample in 112 cases of ovarian neoplasm (Benign ???? 73, Malignant ???? 39). Serum level of CA-125 was significantly (p=0.0001) raised in malignant cases compared to benign neoplasm. The sensitivity and specificity of serum CA-125 was 92.3% and 87.6% respectively. In our study 92.3% of malignant tumour cases had raised level of CA-125.Serum salivary amylase was significantly (p=0.0001) more among malignant tumours (refer table-5). Conclusion: To conclude, in the present study there was significant increase of both serum CA-125 and salivary amylase level in malignant tumour cases than that of benign tumour cases.
Keywords: Ovarian neoplasm, serum CA-125, salivary amylase, histology grade
Full Text:
INTRODUCTION
Ovarian cancer is the second most common of all genital cancers and accounts for 10-15 % of all gynaecological cancers in India and other developing countries 1 . Up till today there is no regular screening test for diagnosing ovarian cancer and most of the neoplasm patient remain asymptomatic; as a result about 80% of malignant ovarian neoplasm are in stage 3 and stage 4 when diagnosed on histopathological examination of tumour tissues after surgery2 . Ovarian neoplasm may develop from its surface epithelium or from germ cell or from stroma of ovary, including the sex cords which are fore runner of the endocrine apparatus of the postnatal ovary. Malignant transformation of ovarian neoplasm is associated with biochemical changes of some parameters. A number of tumour markers is have been used for investigating ovarian cancer.3 Like serum Cancer antigen-125 (CA-125), a non conventional analyte like salivary amylase can be used to screen the ovarian cancers. .There are different studies showing raised levels of salivary amylase in ovarian malignancy. Luca F et al. shown in their study that after ruling out pancreatic and salivary gland disorders if the serum amylase is raised it is indicative of ovarian tumour and therefore can be used as a marker for ovarian cancer4 . Zakowski J J et al. mentioned in 1984 in the Journal of Clinical Chemistry that human serous-type ovarian tumours contain an acidic isoenzyme of amylase. Purified amylase isoenzyme from a human serous ovarian tumour was characterized and compared with the purified salivary and pancreatic isoenzymes. It was found that, ovarian tumour amylase was similar to the salivary and it differs from the pancreatic enzyme by apparent molecular mass and doublet formation on sodium dodecyl sulfate--polyacrylamide electrophoresis (SDSPAGE), specific activity of pure enzyme, and sensitivity to specific alpha-amylase inhibitors. The tumour amylase is clearly distinct from the pancreatic and differs from the salivary enzyme only in net electrical charge otherwise it is similar to or identical with salivary amylase as an enzyme5 . It also reported that alteration in the values of salivary amylase and CA-125 does not occur in 100% of cancer patients. In that case if these two parameters are used it will be helpful for diagnosis and assessment of grades of tumour as well as prognosis in a very simple and economic way. With this view, aim of our present study was to compare serum CA-125 and Salivary amylase level with benign and malignant tumour groups.
MATERIALS AND METHODS
The cross sectional descriptive observational study was conducted in the department of Biochemistry and Pathology, R.G.Kar Medical College and Hospital, Kolkata ,West Bengal, India. The study was undertaken after approval from institutional ethics committee .All the patients were properly counselled and their written consent was taken in the presence of a witness.
Study Group: 112 cases of USG diagnosed women with ovarian neoplasm in the age bracket 15 to 60 years admitted to Gynaecology ward for operation in one year (august 2011 to July 2012) were the study population.
Exclusion Criteria: One who has received radiotherapy or chemotherapy in ovarian neoplasm was excluded from the study. Also excluded were pregnant women, patient with metastatic neoplasm in ovary, women with any other malignancy, pelvic inflammatory disease, endometriosis, patient having salivary disease and patient with history of pancreatitis.
Specimen Collection and Preparation: For accurate comparison, a fasting venous blood sample was taken as specimen and the usual precaution in collecting of venipuncture samples were observed. Three ml of venous blood was collected in a plain vial .Blood was allowed to clot and centrifuzed to separate the serum from cells. Fresh sera was used to measure salivary amylase, rest of the sera was stored in (-20 0 c) in alliquotes before analysis of CA-125. The tests were performed with available instrumen and reagent in the department of Biochemistry. Estimation of serum CA-125:
Estimation of serum CA-125: Estimation of serum CA-125 level by Enzyme linked immune sorbent assay (ELISA) method 6,7. Instrument used was ELISA reader with automatic washer of Robonik-Read well touch.
Estimation of Serum Salivary Amylase: Salivary amylase was determined by subtracting the serum value of pancreatic amylase from alpha (α) amylase i.e. from total amylase value of each serum sample. Serum alpha (α) amylase and pancreatic amylase was analysed using kinetic method8,9 by Semi auto analyzer of Accurex-112 plus .
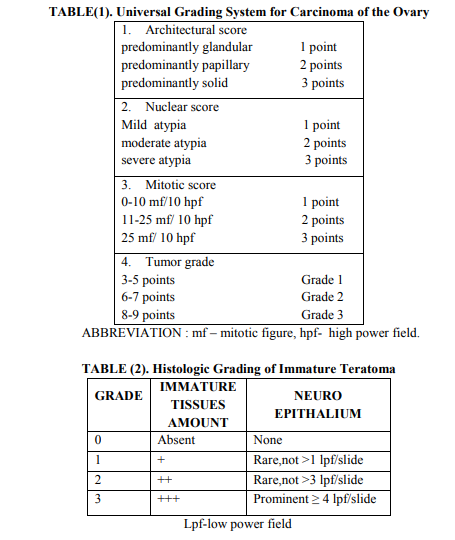
Procedure of Histological Slide Preparation: After operation the specimen was sent for histological examination in the dept Pathology and they were differentiated as benign and malignant tumours. After block preparation (tissue processing) tissue section was cut with the help of microtome (LeicaRM2125 RT) into ribbon like section of 5 microns thickness and floated in warm water (≈ 56 degree C) for expansion of curled section. These sections were than collected on albumenized slides and allowed to dry and fix on slides. Staining was done with Harris Haematoxylin and Eosin stain .Slides were mounted in DPX with coverslip and was examined under microscope (Nuclei - blue/black, Cytoplasm - varying shades of pink or clear. Muscle fibers - deep pink/ red, Red blood cellorange/ red, Fibrin – deep pink.) The universal grading system for all types of ovarian carcinomas proposed by Silverberg and colleagues is widely used10. With this system, the grade of carcinoma was determined by the degree of nuclear atypia, the frequency of mitotic figures, and the extent at which the tumour cells form papillae or glands11,12. Universal grading system is given in table (1). Immature teratomas are graded using a system that rates them from grade 0 for a neoplasm composed of entirely of mature tissues to grade 3 for a neoplasm containing abundant immature tissue .Histologic grading of immature teratoma is dependent on amount of immature tissues present in the tumour 13.Grading system of immature teratoma proposed by Norris H J et al. is mentioned in table-2. All data were analyzed on SPSS (statistical package and subjected to Students). Validity statistics and ROC curve were done. Level of significance was assumed at p value < 0.05.
RESULTS
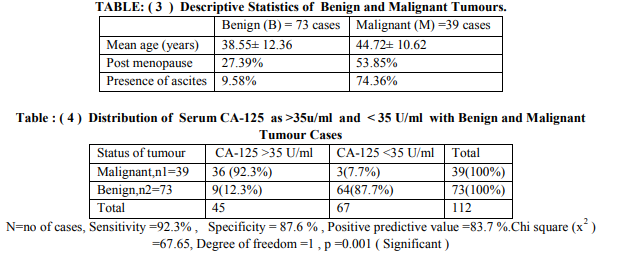
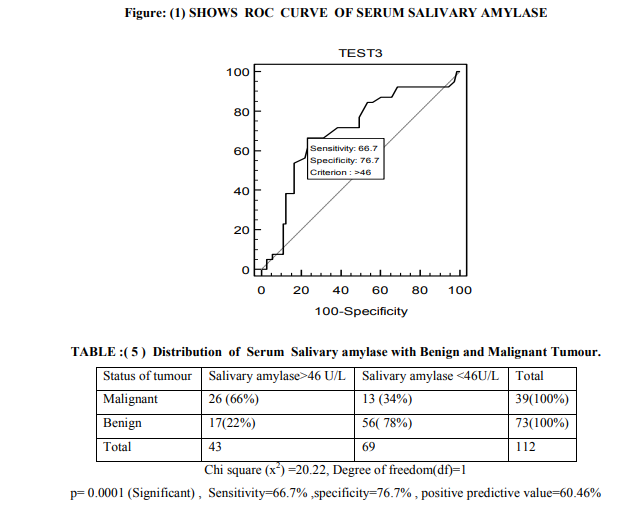
In our study among 112 cases 73 were benign and 39 malignant tumour patients .Mean age of benign neoplasm group and malignant group was 38.55 ±12.36 years and 44.72 ±10.62 years respectively. Out of 39 malignant patients 21 (53.85%) were in post menopause and 29(74.36%) having ascites (refer table - 3). In this study 92.3% of malignant cases had serum level of CA-125 more than 35 U/ml and only 12.3% benign cases had raised level of CA125.CA-125 had sensitivity of 92.3% ,specificity of 87.6% and positive predictive value was 83.7% (refer table-4). According to Youden index (0.4338) cut off level of salivary amylase level was 46 U/L and area under curve was 0.701 (refer to figure-1). With 46U/L cut off sensitivity and specificity were 66.7% and 76.7% respectively. On pre operative serum estimation, 66% of malignant tumour cases had raised level (>46 U/L) of salivary amylase and it showed significant (p=0.0001) difference with benign tumours (refer table-5).
DISCUSSION
There are different case reports showing raised serum level of amylase in malignant ovarian tumour patient mainly due to salivary isoenzyme fraction, but no established reference range is available. In this study pre operative serum level of salivary amylase was estimated to determine its significance in benign and malignant ovarian neoplasm cases. Pre operative serum CA-125 was estimated in study cases to determine its significance in malignant cases, so that only by estimating pre operative serum levels of these two parameters we can predict about status of ovarian tumour patient suffering from and we can plan for early intervention for better prognosis. In the present study 73(65%) were benign and 39 (35%) were malignant cases. Among them 9(12.3 %) patient of benign tumours had elevated level of CA-125 ( >35U/ml to 121 U/ml) whereas 64 ( 87.7 % ) had normal value (<35U/ml) considering cut off value 35U/ml as kit literature6,7. Among malignant tumour, 36 (92.3%) cases had raised CA-125 value and only 3(7.7%) had normal value (refer to table-4). Sensitivity and specificity of serum CA-125 was 92.3% and 87.6% respectively (table-4) .Significant difference (p= 0.001) was found between serum CA-125 and type of ovarian neoplasm. Positive predictive value (PPV) of CA125 in our study was 83.7%. This agrees with the observation of Bast RC et al. They had described that only 1 per cent of 888 apparently healthy persons and 6 per cent of 143 patients with non malignant disease had serum CA-125 levels above 35 U/ml. In contrast, 83 of 101 patients (82 per cent) with surgically demonstrated ovarian carcinoma had elevated levels of cancer antigen (CA-125). Kenemans P et al. also agrees with this observation 14,15 . Receiver operating characteristics (ROC) curve was plotted as there was no reference range for serum level of salivary amylase .According to ROC curve of serum salivary amylase cut off was taken as 46U/L . 26 (66%) malignant tumour cases had serum salivary amylase >46 U/L and 13 (34%) cases had less than 46 U/L (refer table-6). Among 73 benign tumour cases 17 (23.28%) cases had > 46 U/L and 56 (76.72%) cases had < 46U/L of serum salivary amylase .Chi square test shows significant (p=0.0001) difference between benign and malignant cases(refer table-5). From this data analysis we can say that if a patient presenting with ovarian lump has serum salivary amylase >46U/L ,she is more likely suffering from malignancy. But before considering this cut off for screening further study is indicated on a larger population . Each laboratory should establish its own cut off value for serum Salivary amylase. There are case reports showing presence of salivary amylase secreting cells in the tumour tissues. Pose Reino A et al. reported a case of hyperamylasemia produced outside the pancreas. In an ovarian carcinoma patient blood level of amylase was slightly increased16.In another case report an amylase-producing ovarian cancer in 69- year-old woman has been investigated by light and electron microscopy, as well as by amylase isoenzyme analysis. Serum and urinary amylase levels were found to be elevated, especially by an amylase isoenzyme analysis of the tumour homogenate; salivary type amylase was identified in tumour tissue. On amylase isoenzyme electrophoresis they detected the salivary isoenzyme in ascitic fluid17.But in this study the sample size is not large enough to draw such conclusion, future study is indicated on large population to detect the significant relation between malignant tumour and serum salivary amylase.
CONCLUSION
To conclude, in the present study there was significant increase of both serum CA-125 and salivary amylase level in malignant tumour cases than that of benign tumour cases. Moreover due to ease of testing and cost effectiveness, serum salivary amylase can be used alone or as an adjunct to CA-125 in screening and monitoring patients of Ovarian neoplasm especially in remote areas where sophisticated facilities are not available.
ACKNOWLEDGEMENT
We want to acknowledge Prof. (Dr.) Ajit Ranjan Bhattacharyya, HOD, GandO department of R.G Kar Medical College and Dr. Rupam Karmakar, Associate Professor, Department of pathology of R.G Kar Medical College for their co-operation during the whole course of work.
CONFLICT OF INTEREST: None.
References:
1. Padubidri V G, Daftary SN. Disorders of ovary.In: Shaws Text book of Gynaecology. 13th edn.Lajpat Nagar, New Delhi: Elsevier; 2004, 354-372.
2. Padubidri VG, Daftary SN. Disorders of ovary.In: Shaws Text book of Gynaecology. 13th edn.Lajpat Nagar, New Delhi: Elsevier; 2004,397-400.
3. Joel Larma,Ginger J. Gardner, Ovarian Cancer.in Fortner, Kimberly B.; Szymanski, Linda M.; Fox, Harold E. et al. 3rd Edition .Johns Hopkins Manual of Gynecology and Obstetrics, Baltimore, Maryland: Lippincott Williams and Wilkins,2007:510-525
4. Luca Frulloni, Franca Patrizi, Laura Bernardoni, Giorgio Cavallini. Pancreatic Hyperenzymemia: clinical significance and diagnostic Approach. JOP. J. Pancreas.2005;6(6):536-551.
5. Zakowski J J, Gregory M R and Bruns D E.Amylase from human serous ovarian tumors: purification and characterization. The American Association for Clinical Chemistry, 1984 ; 30: (1), 62-68
6. Hasholzner U,Steiber P, Baumgartner L ,et al.Clinical significance of tumour marker CA125 ? and CA-72-4 in Ovarian carcinoma,Int journal Cancer,1996;69:329-334
7. Sikorska H,Schuster J, Gold P.Clinical application of Cancer antigen 125,Camcer detection preview,1988;12:321-355
8. IFCC Methods for measurement of catalytic concentrations of enzymes,J.Clin.Chemistry.Acta.1999;281:5
9. David H.Hydrolysis by human alpha amylase of p-nitrophenyl oligosaccharides containing four to seven glucose unit.Clinical Chemistry.1982;28:1485-89
10. Silverberg S G. Histopathologic Grading of Ovarian Carcinoma:a review and proposal.International Journal of Gynaecol Pathology,2000;19:7-15.
11. Shimizu Y,Kamoi S,Amada S et al.Toward the development of a universal grading system for ovarian epithelial carcinoma-Testing of a proposed system in a series of 461 patient with uniform treatment and followup.Cancer,1998;82:893-901
12. Shimizu Y,Kamoi S,Amada S et al.Toward the development of a universal grading system for ovarian epithelial carcinoma.I.Prognostic Significance of histologic features-Problems involved in tha architectural grading system.Journal of Gynaecological Oncology.1998;70:2-12
13. Norris H J, Zirkin H J, Benson W L.Immature (Malignant) teratoma of the ovary.A clinical and Pathologic study of 58 cases.Cancer,1976,37:2359-2372
14. Bast RC, Jr, Klug TL, St John E, Jenison E, Niloff JM et al. A radioimmunoassay using a monoclonal antibody to monitor the course of epithelial ovarian cancer. N Engl J Med. 1983;309:883–887
15. Kenemans P, van Kamp GJ, Oehr P, Verstraeten RA. Heterologous doubledeterminant immunoradiometric assay CA 125 II: reliable second-generation immunoassay for determining CA 125 in serum. Clin Chem. 1993;39:2509–2513
16. Tanaka Y, Ueda G, Tanizawa O, Miyake H, Kohno M, Nishiyama S et al.A case of amylase-producing ovarian tumor. Gan No Rinsho. 1986 Jan;32(1):111
17. Kawamurra S, Hiura M, Kaneko H, Yokoyama T, Tanaka M, Chiba T.Amylaseproducing ovarian cancer treated with postoperative QF chemotherapy--a case report. Gan No Rinsho. 1988 Apr;34(4):497-504.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License