IJCRR - 5(21), November, 2013
Pages: 70-77
Date of Publication: 21-Nov-2013
Print Article
Download XML Download PDF
HISTOPATHOLOGICAL PATTERN OF OVARIAN NEOPLASMS AND ESTROGEN AND PROGESTERONE RECEPTOR EXPRESSION IN PRIMARY EPITHELIAL TUMOURS AND THEIR HISTOPATHOLOGICAL CORRELATION
Author: Summyia Farooq, Reyaz Tasleem, Naila Nazir, Ruby Reshi, Zhahid Hassan
Category: Healthcare
Abstract:Background: Ovarian neoplasm is one of the most complex tumours of women in terms of histogenesis, clinical behaviour and malignant potentiality and is the third most common cancer of female genital tract. Estrogen and progesterone hormones and their receptors are thought to be involved in process of tumour genesis in ovarian cancers. The objective of this study was to evaluate histopathology of ovarian neoplasms and evaluate the expression of estrogen and progesterone receptors in primary epithelial tumours. Material and Methods: The study included a total of 144 consecutive cases of ovarian tumours. Sections of 4\?m from 50 epithelial tumours were taken from paraffin embedded tissue blocks for immunohistochemistry (IHC) for ER and PR. Statistical analysis was done using chi square test. Results: Among 144 cases 75% were benign 3.48% were borderline and 21.52% were malignant. Surface epithelial tumours were most common 69.44% followed by germ cell tumours 22.22%. ER and PR had higher expression in serous tumours 37.14% and 51.42% as compared to mucinous tumours 6.67% and 13.3%. ER and PR had lower expression in benign tumours 20% and 30% as compared to borderline 50% and 50% and malignant 61.53% and 84.6%. ER and PR had higher expression in older age women 78.57% and 75%. ER and PR expression in grade 3 tumours was 75% and 87% and in advanced stage was 100% each. Conclusion: Surface epithelial tumours were most common type of tumours both benign as well as malignant. Estrogen and Progesterone showed higher expression in serous tumours, older age group and higher grade of tumours. So estimation of Estrogen and Progesterone receptor status may help to select the women with ovarian malignancy for hormonal therapy, which is more likely to improve the response rate as well as prognosis.
Keywords: Ovarian tumours, Molecular profile, ER, PR, Histopathology.
Full Text:
INTRODUCTION
Ovarian tumours are one of the major health problems and the most complex tumours of women and are the third most common cancer of the female genital tract1,16. Ovarian cancers account for 15.0% - 25.0% of all primary malignancies in female genital tract and are common cause of death from gynaecological malignancies1. It is estimated that about 1 in every 70 women have a life time risk of developing ovarian cancer18. The most significant risk factor for developing ovarian cancer is age. Ovulation, growth factors, cytokines, and environmental agents may play a role in the initiation and progression of ovarian cancer22. Familial predisposition has been noticed in approximately
5-10% of cases6,10. The risk for developing ovarian cancer is increased four fold in women with affected first degree relative. Estrogen and progesterone are important hormones secreted by ovary acting through specific receptors19. Both the hormones and their receptors are thought to be involved in the process of tumour genesis in ovarian cancers14. Immunohistochemical ER and PR assays have added the advantage that the distribution in tumour tissues as well as normal surrounding can be evaluated9. Hormone receptor determination in malignant ovarian neoplasms can aid in selection of patients for endocrine therapy in a manner similar to that already established for certain hormone dependent cancers5.
The present study is an effort addressing the frequency of ovarian tumours, their histological subtypes and hormonal status in epithelial ovarian tumours observed over a period of two years in our valley. The study also aims at studying the correlation between histopathology and hormonal receptor status (ER and PR status), so that patients can be subjected to hormonal therapy.
MATERIAL AND METHODS
After obtaining the ethical board approval, a two year prospective study was carried out from January 2011 to February 2013 on 144 patients in the post-graduate department of pathology, Government medical college Srinagar. All ovarian neoplasms except functional ovarian cysts received in department were included in study. Complete clinical data of patients was recorded. The received respected specimen was put in 10% formalin for fixation overnight. The specimen was then grossed findings like appearance, size were recorded and multiple tissue pieces from representative areas 1 cm apart were taken with a knife, put in stainless steel cassettes, labelled and again kept in fixative for 2-4 hours. The tissue blocks were thoroughly washed with distilled water three to four times to remove excess formalin. The tissue was then dehydrated by passing through ascending grades of ethanol and the tissue was then embedded in molten wax, which was maintained in an oven at melting point of wax. Wax blocks of embedded material were prepared by using Leukhart’s ‘L’ blocks, these blocks were marked and labelled for further processing. Then the sectioning of tissue blocks was done by rotatory microtome and 3-5 micron sections were obtained. A thin film of Mayer’s albumin was spread over clean glass slides and sections were placed on these slides using hot water bath. After de-waxing the staining of sections was done routinely with Haemtoxylin and Eosin. Then slides were studied and tumours were classified according to WHO classification. Histological subtypes, differentiation (grade) of malignant tumours were studied. Immunohistochemical expression for ER and PR was studied in 50 epithelial tumours which included all malignant (20), borderline (5) and 25 randomly selected (table of random numbers) benign epithelial tumours. Immunohistochemical staining was done using rabbit monoclonal anti-human estrogen receptor antibody (clone SP1Biocare) and anti-human progesterone receptor antibody (clone SP2 Biocare) by microwave antigen retrieval technique, in a standard avidin- biotin immunoperoxidase method. Diaminobenzidine (DAB) with hydrogen peroxide was used as chromogen substrate and sections were counterstained by Mayer’s Haematoxylin. For immunohistochemical staining, positives control sections were from breast carcinoma and negative controls sections were provided by omission of primary antibody. Grading of ER and PR staining was performed using H-scoring system obtained by multiplying the epithelial cell intensity ( graded as 0 negative, 1+ weak, 2+ as moderate and 3+ as strong) by percentage of positive cells. This index ranges from 0-300; 300 indicates, 100% of tumour cells stained strongly (3+). H-score of 0-9 is taken as negative and > 9 as positive. Statistical analysis was done using Chi Square test. SPSS software was used for data analysis.
RESULTS
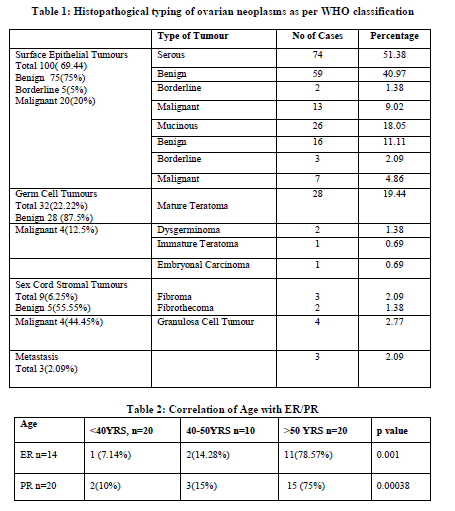
The age of the patients ranged from 15 – 70 years with an average of 43.6 years. Among 144 cases 108 (75%) were benign, 5 (3.48%) were borderline and 31(21.52%) were malignant. In present study out of 144 cases 131(90.97%) cases were unilateral and 13(9.03%) were bilateral. Adopting WHO classification the surface epithelial tumours were maximum accounting to 100 (69.44%) cases followed by germ cell tumours 32 (22.22%), sexcord stromal tumours 9(6.25%) and metastatic tumours 3(2.09%). The surface epithelial tumours were maximum (100) cases out of them 75 (75%) were benign 5 (5%) borderline and 20 (20%) were malignant (Table 1).
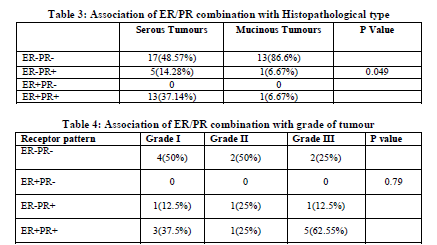
Out of 144, IHC for ER and PR was done on 50 primary epithelial tumours, which included 20 primary malignant 5 borderline and 25 randomly selected benign tumours. Out of 50 epithelial tumours estrogen positivity was seen in 14 (28%) and progesterone positivity was seen in 20 (40%) of cases. Out of 14 ER positive cases 13 were serous tumours and only one was mucinous. Out of 20 PR positive cases 18 were serous and two were mucinous. Out of 35 serous tumours estrogen receptors were expressed in 13 (37.14%) and progesterone receptors were expressed in 18 (51.42%) cases. Out of 15 mucinous tumours estrogen receptor positivity was seen in only 1(6.67%) and progesterone receptor positivity was seen in 2 (13.3%) cases. Out of 35 serous tumours 13(37.14%) were both ER PR positive and 17(48.57%) were both receptors negative. Out of 15 mucinous, only one (6.67%) was both receptors positive and 13 (86.6%) were both receptor negative. So both ER and PR showed statistically significant higher expression in serous tumours (37.15%/ 51.42%) as compared to mucinous tumours 6.67% /13. In serous tumours estrogen receptors were expressed in 8 (61.53%) malignant tumours. 1( 50%) borderline and 4 (20%) of benign tumours, and progesterone receptors were expressed in 11 (84.6%) of malignant 1 (50%) borderline and 6 (20%) benign tumours. In mucinous tumours estrogen receptors were expressed in only 1(14.28%) malignant tumour and progesterone receptors were expressed in 1(33.3%) borderline and 1(14.28%) malignant tumour. Among 35 serous tumours 17 (48.57%) tumours were both ER/PR-, none was ER+/PR-, 5 (14.28%) were ER-/PR+ and 13 (37.14%) were both ER+/PR+. Among 15 mucinous tumours 13 (86.6%) were both ER-/PR-, 1(6.67%) was ER+/PR+ and 1(6.67%) was ER-/PR+. Table 2, 3 and 4 depict the correlation between the age, histological type and tumour grade with ER/PR status respectively.
DISCUSSION
Ovarian neoplasm is the most fascinating tumour of women in terms of histogenesis, clinical behaviour and malignant potentiality. Many of ovarian neoplasms cannot be detected early in their development, they account for a disproportionate number of fatal cancers, being responsible for almost half of deaths from cancer of female genital tract4. The present study was carried out on 144 cases of ovarian neoplasms. Two main aspects considered in the study were histopathological type as per WHO classification and estrogen and progesterone receptors expression in primary epithelial ovarian neoplasms and their correlation with histopathology, age and differentiation (grade) of tumour.
The maximum number of tumour occurred in 31-40 years 50(34.73%) followed by 21-30 years 45 (31.25%), 41-50 years 22 (15.28%). Similar age range was reported by other studies7,15,21.
The epithelial tumours on which immunohistochemistry for ER and PR was done, 35 were serous and 15 mucinous tumours as no other epithelial tumour type was found in present study. Estrogen positivity was seen in 14 cases (28%) and Progesterone receptor positivity was seen in 20 cases (40%) of 50 epithelial tumours. Scambia et al (1998)5 reported that 56% and 35% cases of ovarian cancers were positive for ER and PR respectively. Tangjitgamol et al (2009)20 in their
study found that ER positivity was 39.6% and PR positivity was 33.0% of total 106 cases of ovarian tumours. Both ER and PR showed higher expression in serous tumours 37.14% and 51.42% as compared to mucinous tumours 6.67% and 13.4%. H. Arias-pulido et al (2009)8 reported that 66% of serous tumours were ER positive and 54% were PR positive, in mucinous tumours 13% were ER positive and 20% were PR positive. Sylvia et al (2012)11 showed 72% of serous tumours were ER positive and 57.7% were PR positive, Mucinous tumours were negative for ER and 14.28% were positive for PR In present Estrogen receptor showed higher expression in malignant tumours 61.53% as compared to benign (20%) and borderline 50%. In present study Progesterone receptor expression was 30% in benign, 50% in borderline and 81% of malignant tumours. Our observations were similar to Agarwal et al13 who reported progesterone receptor positivity in 8 (88.9%) in malignant tumours but no receptor positivity was seen in benign tumours but in present study PR positivity in benign tumours is 30%, which is in close to Sylvia et al11 who showed 40% benign tumours expressed PR. In present study out of 14 ER positive cases only 1 (7.14%) was < 40 years, 2 (14.28%) were between 40-50 years and 11(78.57%) were more than 50 years of age. Out of 20 PR positive cases 2 (10%) were <40 years 3(15%) were between 40-50 years and 15(75%) were >50 years of age. Our observations are similar to Sylvia et al11, Hahnel et al17 and Agarwal et al13. In present study Estrogen receptors were mostly expressed in poorly differentiated (grade III) and moderately differentiated 50% each as compared to well differentiated 37.5% and progesterone receptors were expressed as 62.5% in well differentiated 50% in moderately differentiated and 62.5% in poorly differentiated. Our study is in contrast to Buchynska et al (2009)3 who demonstrated that higher grade tumours have low estrogen receptor positivity. Lau et al (1999) demonstrated that progesterone receptor decreases with increasing grade this is in contrast to our observation. We could not find significant association between PR positivity and grade. In present study ER was expressed as 100% in stage IV, 50% in stage III, 50%in stage II and 30% in stage III. PR was expressed as 50% in stage I, 50% in stage II, 75% in stage III and 100% in stage IV tumours. Hogdall E V et al (2007)12 reported that ER positivity in ovarian cancers increases with increasing FIGO stage. Agarwalet al13 reported that receptor positivity increases with stage of tumour. Similar observations were made by Sylvia et al11. Slotman et al2 reported 64% and 60% of stage III and IV were positive for ER and PR respectively as compared to 43.7% and 56% of stage I and II tumours.
CONCLUSION
In the current study benign tumours were more common than malignant tumours in all age groups. Estrogen and Progesterone showed higher expression in serous tumours, older age group, higher grade and in advance stage of tumour. So estimation of Estrogen and Progesterone may help to select the women with ovarian malignancy for hormonal therapy, which is more likely to improve the response rate as well as prognosis.
ACKNOWLEDGEMENTS
We thank Dr. Sheikh Bilal, Dr. Ambreen and Dr. Mahnaaz for helping us in compiling the data.

References:
- Benson RC, Diagnosis and treatment, current obstetrics gynaecol 1976, 1:236.
- BerendJ. Slotman., RadkaKuhnel., B Ramanath Rao., Gerard. H., Jan De Graaff and Johannes. Importance of steroid receptors and aromatase activity in the prognosis of ovarian cancer: High tumour progesterone receptor levels correlate with longer survival. Gynaecologic oncology 33,76-81(1988).
- Buchynska LG, Iurchenko NP, Grinkevych VM, Nesina IP, Chekhun SV, Svintsitsky VS. Expression of the estrogen and progesterone receptors as prognostic factor in serous ovarian cancers. ExpOncol 2009; 31:48-51.
- Crum CP. The female Genital tract. In: Kumar V, Abbas AK Fausto N. editors. Robbins and Cotran Pathologic Basis of Disease. 7th ed. Philadelphia: W.B Saunders. 2004;1059-1118.
- G scambia, G Ferrandina, G D Agostino, A Fagotti, M DI Stefano, F Fanfani, F G Serri and S Mancuso. Oestrogen and progesterone receptors in ovarian cancer. Endocrine-Related cancer 1998,5;293-301.
- Greggi S, Genuardi M, Benedetti-Panici P, Cento R, Scambia C, Neri G, Mancuso S. Analysis of138 consecutive ovarian cancer patients. Incidence and characteristics of familial cases. Gynaecoloncol 1990, 39: 300-304.
- Gupta SC, Singh PA, Mehrotra TN, Agarwal R. A clinico-pathological study of ovarian tumoursindian J Pathol Microbiol.1986 Oct;29(4):354-62.
- Hugo Arias-Pulido, Harriet O. Smith, Nancy E.Joste, Therese Bocklage, Clifford R. Qualis, Allison Chavez, Eric R. Prossnitz, Claire F, Verschraegen. Estrogen and progesterone receptor status and outcome in epithelial ovarian cancers and low malignant potential tumour. Gynaeologic oncology 114;2009:480-485.
- IsolaJ, Kallioniem OP, Korte JM, Walstrom T, Anie R et all 1990 steroid receptors and Ki 67 reactivity in ovarian cancer in normal ovary, correlation with DNA flow cytometry biochemical receptor assay and patient survival.
- Lynch HT, Watson P, Bewtra C, Conway TA, Hippee CR, Kaur P Lynch JF, Ponder BA. Hereditary ovarian cancer. Cancer 1991,67 1460-1466.
- Mary T Sylvia, Surendra Kumar, Papa Dasari The expression of immunohistochemical markers estrogen receptor, progesterone receptor, Her-2-neu, p53 and Ki-67 in epithelial ovarian tumours and its correlation with clinicopathologic variables Indian J PatholMicrobiol.2012 ,55(1); 33-37.
- MR. Cardillo. E. Petrangeti. N. Aliotta. L Salvatorv, L Ravenna1, C. Chang*, C. Casiagna Androgen Receptors in Ovarian Tumours: Correlation with Oestrogen and Progesterone Receptors in an immunohistochemical and Semi quantitative Image Analysis Study. J. Exp. Clin. Cancer Rev. 17.2, 1998.
- N.Agarwal, D.L Rao, K. Murgeshan, U Verma, S. Mittal and T.N Chapekar. Clinical evaluation of steroid receptors in ovarian neoplasms. Int. J. Gynaecol. Obstet. 1987,25 145-149.
- Pertschuk LP, Beddee AM, Gorelic LS, Shain SA immune cytochemicalassay of estrogen receptor in endometroidcarcinoma with monoclonal antibodies Cancer 1986;57:1000-1004.
- Rajagopalan K, Rajagopalan C.K andGourikutty A.K: Ovarian tumours a clincopathological study. Ind J Path Microb.1982 79; 73-75.
- Robbins and Cotran Kumar et al: Ovaries ch.22 pathologic basis of disease 8th ed. Elsevier 2010.
- Roland Hahnel, Dr. Rer. George R.H. Kelsall, John D. Martin, Anthony JT et al Estrogen and progesterone receptors,tumors of human ovary Gynecologic oncology 13,145-151(1982).
- Schiff M. Becker TM. Smith HO. Gilliland FD. Key CR et al. Ovarian cancer incidence and mortality in American Indian, Hispanic, and non-Hispanic white women in New Mexico. Cancer Epidemiology, Biomarkers andPrevention. 1996 May;5(5):323-7.
- Shabani N, Mylonas I, Jeschke U, Thaqi A, Kuhn C, Puncher T, .Expression of estrogen receptor alpha and bete and progesterone receptors A andBin human mucinous carcinoma of endometrium ,(Anticancer Res 2007 .27 (4A):2027-33.
- Tangjitgamol S, Manusirivithaya S, Khunnarong J, Jesadapatarakul S, Tanwanich S Expressions of estrogen and progesterone receptors in epithelial ovarian cancer: a clinicopathologic study Int J Gynecol Cancer. 2009 May; 19(4):620-7.
- Tyagi SP, Madan A, Mohsin S, Hameed F, Saxena K. Epithelial tumours of the ovary Indian J Pathol Microbiol. 1978 Oct; 21(4):281-9.
- Vanderhyden, B. C., Shaw, T. J Garson, K., Tonary, A. M. 2003. Ovariancarcinogenesis.
|






 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License